Research
The Hohloch Group focuses on the use of early transition and f-Block metals in sustainable and environmentally benign applications. These span from their use in molecular magnetism, sustainable chemistry catalysis or to activate inert small molecules such as N2, CO2, H2 and heavy cyanates of the general formula [ChCPn]- (Ch = Chalcogen, O, S, Se and Pn = Pnictogen, N, P, As). The aim is to convert them into value-added chemicals and basic feedstock materials for chemical industries.
In this context, our research focusses mainly on the use of two "non-classical" ligand classes in the regime of these electropositive metals: N-heterocyclic and mesoionic carbene (NHC and MIC) and phosphine donors. Both these ligands classes are strong donor ligands, which have been barely explored in the chemistry of the early transition metals and the f-Block elements and have been proposed to only show weak interactions with the metals in question. To ensure the binding of the ligands in question, anionc tether groups (phenolates, alcoholates or amides) are attached, forcing a stronger metal-carbene / phosphine interaction. This results in the formation of new metal complexes with intruiging reactivities supported by the strongly donating ligand.
In addition to the synthesis of such novel and highly reactiv metal complexes, we focus on the cutting-edge spectroscopic exploration of these systems. This is done by a varierty of electrochemical methods in combination with spectroscopic techniques such as UV-Vis, IR and EPR spectroscopy as well XAS to further determine the electronic structure and oxidation states of the metal complexes designed. The results obtained are further probed and explored by computational investigations.
In summary, this multi-disciplinary approach helps us to get new insights into the paradigms controlling the reactivity of these complexes and creates future dogmas how the reactivity can be tuned and tailored.
Current research focusses on the following topics:
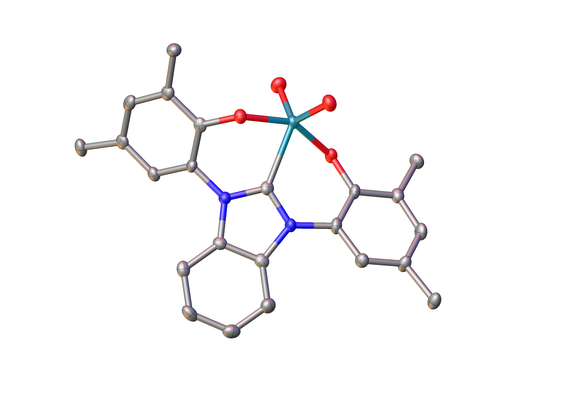
High-valent molybdenum complexes have recently been found to be valuablecatalystsfor deoxygenative transformations, including the reduction of nitroarenes and the deoxygenation of biomass to olefines.
Especially the latter process is crucial to ensure our supply of feedstock chemicals for industry during our shift from fossil fuels to renewable and green energy sources.
We focus on the development of new catalytic systems capable of performing catalytic deoxygenations using N-heterocyclic carbenes as spectator ligands. Our aim is to understand the underlying mechanism and to design new efficient catalysts.
Selected Publications:
Organometallics 2019, 38, 19, 3719–3729
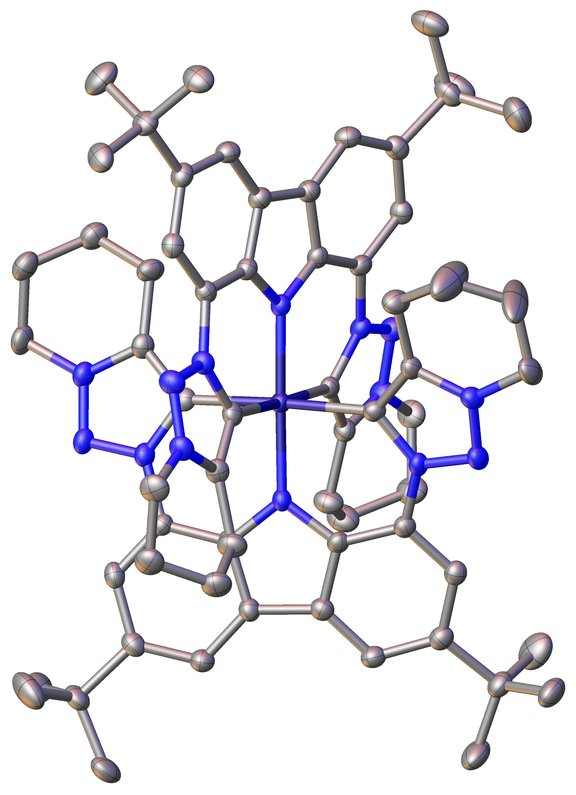
The chemistry of late transition metal NHC / MIC complexes is well explored. Contrary the potential of this unique ligands have been barely touched in the early transition metal regime. Thus, we focus on the use of anionic chelating NHC / MIC ligands to further explore their potential in the chemistry of group V to group VII metals. Special emphasis is thereby put on the redox-variability, finding how many oxidation states these ligands can stabilize / tolerate and the use of these complexes in catalysis (e.g. hydrofunctionalisation, group transfer and polymerisation catalyis).
Selected Publications:
Dalton Trans., 2019,48, 14611-14625
Inorg. Chem. 2021, 60, 20, 15421–15434
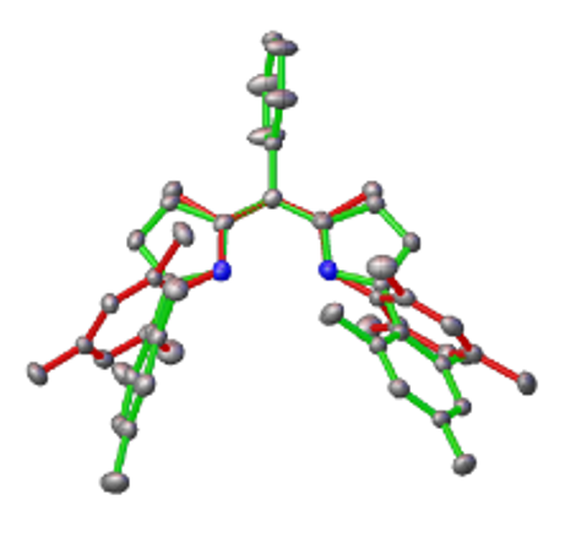
Dipyrrin ligands can be simplified seen as "half-porphyrins", giving them unique electronic and steric properties. Compared to the uniquitously used beta-diketiminate (BDI) ligands, they offer a enhanced steric protection of the metal center, surpressing undesired side reactivites providing a unique potential to study and isolate highly reactive metal complexes. We focus on the stabilization of cationic group IV, V and VI complexes, with the ultimate aim to activate and transfer dihydrogen to organic molecules., with the aim to replace precious metal such as palladium or platinum by earth abundant and environmentally benign metals in catalysis.
Selected Publications:
Inorg. Chem. 2020, 59, 14, 9847–9856
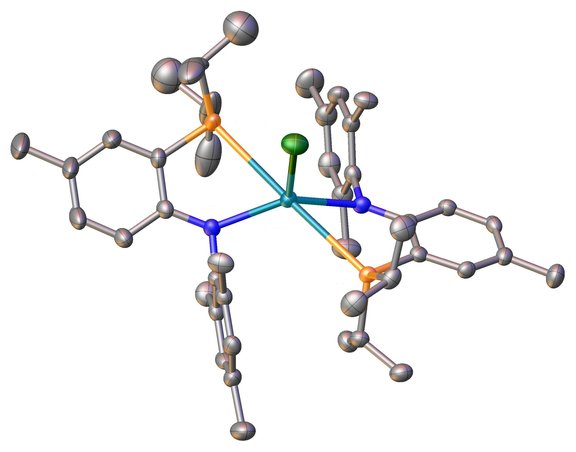
Although the chemistry of lanthanides has been explored for decades, the majority of these studies still focusses on the use of the ubiquitous cyclopentadiene ligand (C5R5) which was among the first ligands used in lanthanide chemistry. However, to develop a rich field in chemistry, also other spectator and non-spectator ligands need to be explored.
One class of ligands that hasbeen well studied in transition metal chemistry, but has only been poorly investigated in f-element chemistry, are phosphine derived ligands with chelating PN(P) coordination motifs. Our goal is to develop new, robust metal complexes using these kind of unique ligands and study their reactivity, their electrochemical and spectroscopic properties.
Selected Publications:
Inorg. Chem. 2020, 59, 5, 2719–2732
Chem. Commun. 2020, 56, 15410-15413