Research
The Dielmann Group advances molecular inorganic chemistry and homogeneous catalysis with a focus on challenges relevant to the energy transition. We design and synthesize reactive main group species and transition metal complexes to achieve energy‑efficient activation and conversion of inert small molecules (e.g., CO2, SF6, H2, N2) and strong chemical bonds, transforming greenhouse gases and renewable resources into value‑added chemicals and materials. Our strategy integrates experiment, spectroscopy, and computation to elucidate bond‑activation mechanisms and guide molecular design. Driven by fundamental discovery, we address pressing scientific challenges through innovative reactivity concepts and new synthetic methods.
A unifying theme is the strategic use of strong substituent effects to predictably tune molecular properties and push them to extreme regimes. We develop strongly donating neutral ligands and ambiphilic main‑group compounds bearing N‑heterocyclic imine (NHI) or N‑heterocyclic olefin (NHO) substituents, including electron‑rich phosphines, low‑coordinate phosphorus motifs, carbenes, pyridines, and phosphonioacetylides. Guided by innovative ligand architectures, we create catalysts based on main group elements and earth‑abundant 3d metals to replace precious‑metal systems and enable new reactions. These advances drive homogeneous catalysis and the development of stimuli-responsive molecular materials.
Superbasic phosphines:
Tertiary phosphines are widely used in many areas of synthetic chemistry with profound impact on organic synthesis, coordination chemistry and catalysis. We aim at expanding the accessible stereoelectronic properties of phosphines beyond their classical boundaries to provide new opportunities in phosphine-mediated transformations and catalysis. As a starting point, we discovered that the electron-donating ability of phosphines can be significantly enhanced by equipping them with strong π-donor substituents such as imidazoline-2-ylidenamino groups. The ability of these substituents to act as π-donor towards phosphorus has a significant influence on the phosphine’s electronic properties. As a consequence, phosphines equipped with three donor groups are ranked amongst the strongest nonionic Brønsted and Lewis bases. The novel phosphines are promising ligands in organometallic catalysis. Moreover, they were used for the reversible complexation and cleavage of CO2, the generation of sulfur monoxide from sulfur dioxide, and the selective fragmentation of SF6.
Selected Publications:
Angew. Chem. Int. Ed. 2015 | Chem. Eur. J. 2017 | Organometallics 2019 | Chem. Eur. J. 2020 | Angew. Chem. Int. Ed. 2022 | Inorg. Chem. 2025, 64, 24134-24142 |Chem. Sci., 2025, 16, 13189-13195
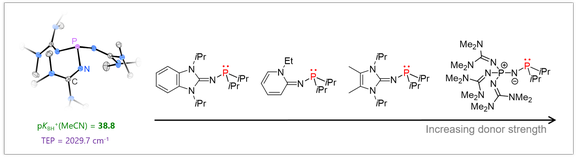
Low-coordinate phosphorus species:
We are interested in the synthesis of novel main group species and explore new concepts to predictable control their molecular properties. By exploiting strong π donor substituents, we stabilized molecular Lewis acids, such as chalcogenophosphonium and phosphorandiylium ions, with the goal to apply them in main group element catalysis and bond heterolysis reactions.
Selected Publications:
Angew. Chem. Int. Ed. 2018 | J. Am. Chem. Soc. 2020 | Nat. Chem. 2019
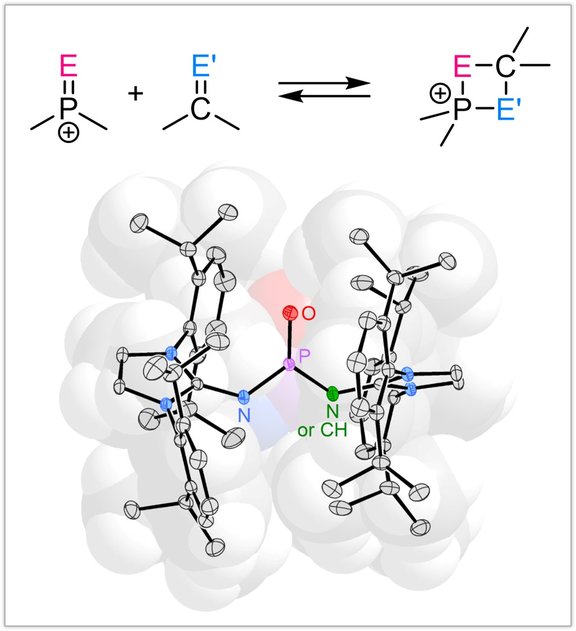
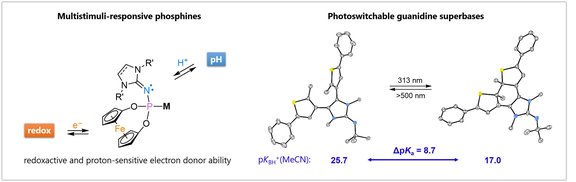
Strongly donating, switchable ligands:
Design, modification and tailoring of ancillary ligands are key tools to tune the activity, productivity and selectivity of homogeneous catalysis. We use stereoelectronically variable N-heterocyclic imine (NHI) substituents to tune the properties of privileged ligand scaffolds.. The basic NHIs are also utilized in the context of metal-ligand cooperation and to introduce stimuli-responsive components into our ligands.
Selected Publications:
Chem. Eur. J. 2021 | Angew. Chem. Int. Ed. 2022 | Adv. Mater. 2024, 36, 2411307
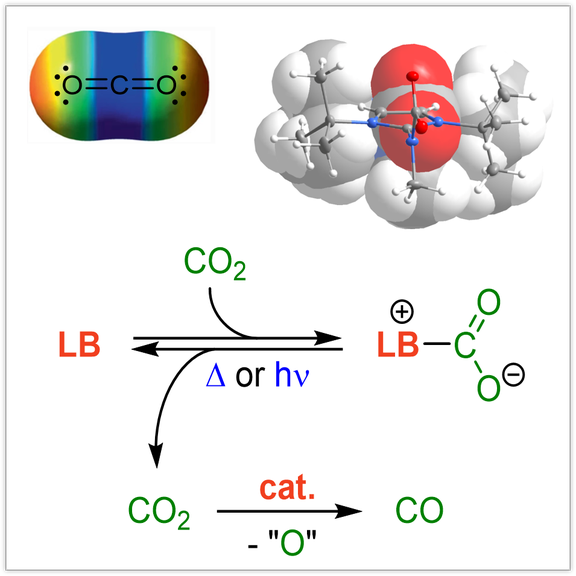
CO2 capture and conversion:
Selective and reversible capture of CO2 either from flue gas or directly from air and its transformation into value-added chemicals, materials and fuels is one of the most important goals to mitigate CO2 accumulation in our atmosphere. In this context, we explore different strategies to activate CO2 in an energy-efficient manner including the formation of reversible Lewis base adducts, composite systems for electrochemical CO2 reduction and photo-triggered CO2 capture-and-release systems. The conversion of CO2 to CO using molecular systems is also under investigation in our laboratories.
Selected Publications:
J. Am. Chem. Soc. 2016 | Green Chem. 2019 | ACS Sustainable Chem. Eng. 2021
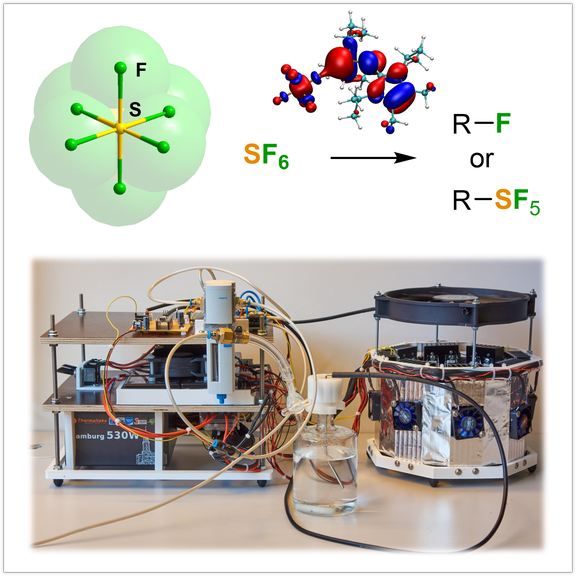
SF6 activation and derivatization:
Sulfur hexafluoride is a fascinating molecule with physicochemical properties that are distinct from that of all other sulfur halides, as well as other gases. It is therefore used in a number of industrial applications and processes, although it is recognized as the most potent greenhouse gas in the atmosphere. Due to the extreme chemical inertness of the octahedral molecule, methods involving the direct fragmentation of SF6 require forcing conditions leading to high energy consumption and the formation of toxic and corrosive products. The chemical valorization of SF6 under mild conditions has therefore garnered considerable attention. Our research interests interest is to develop user-friendly methods for the chemical activation of SF6, which will ultimately open up new avenues for SF6 derivatization.
Selected Publications:
Angew. Chem. Int. Ed. 2018| Angew. Chem. Int. Ed. 2022 | Green Chem. 2022 | Nature Communications | (2026)17:465
Polyurethane catalysts:
Polyurethanes (PUs) represent one of the most important classes of polymer materials with numerous technical applications, including elastomers, rigid foams, soft foams, coatings and adhesives. Organotin compounds are important catalysts in aliphatic polyurethane synthesis due to their high catalytic activity and advantageous properties. Due to their toxicity, however, it has been a long-standing goal to develop more environmentally benign catalysts. In this context, we develop catalysts for PU applications based on main group elements. For instance we showed that the bismuth-catalyzed urethane reaction is significantly accelerated by generating heterobimetallic mixtures of bismuth and lithium carboxylates.
Selected Publications:

Photosensitizers:
Photoactive complexes play a central role in light-enabled chemical processes including solar energy conversion, photoredox catalysis, luminescent devices, and bioimaging. While precious metal systems dominate many photocatalytic applications due to long‑lived excited states and tunable redox properties, we develop photosensitizers from earth‑abundant, low‑cost elements through innovative ligand design, enabling sustainable, scalable photochemistry.
Selected Publications:
Chemistry - A European Journal, 2026; 32:e03127 | Inorg. Chem. Front., 2025, 12, 6663-6675
