Publications
Peer-Reviewed Publications Patents Editorials PhD Thesis
Peer-Reviewed Publications
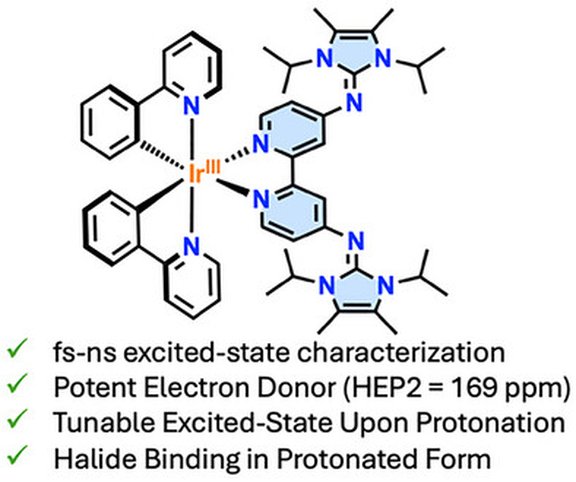
71. pH-Switchable Excited-State Dynamics of Iridium(III) Photosensitizers Bearing an N-Heterocyclic Imine-Functionalized 2,2’-Bipyridine
S. Lempereuur, F. Glaser, J. H. Franzen, L. Schöndorf, M. Seidl, L. Troian-Gautier, F. Dielmann
Chem. Eur. J. (2025), e03127; Link
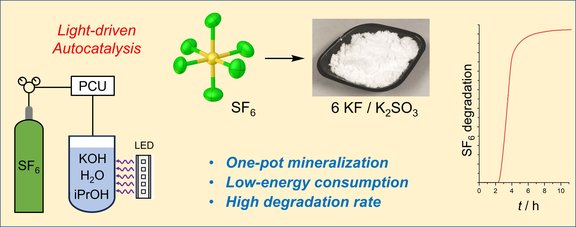
70. Autocatalytic degradation of the extremely potent greenhouse gas SF6 in basic alcoholic solution
A. Sietmann, P. Heinzel, J. Gamper, D. Leitner, L. C. Pasqualini, F. R. S. Purtscher, H. Kopacka, T. S. Hofer, A. Zemann, F. Dielmann
Nat. Commun. (2025); Link
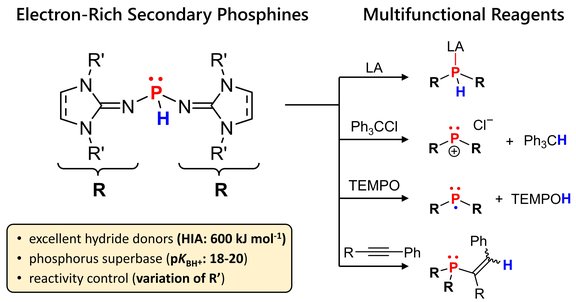
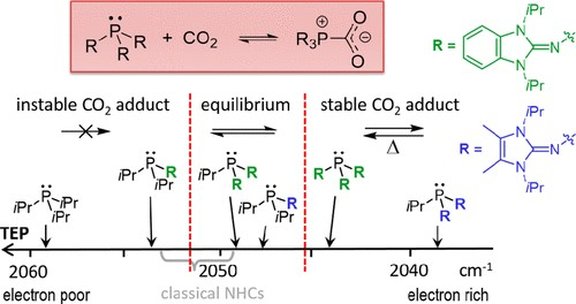
69. Secondary Phosphines Bearing N-Heterocyclic Imine Groups: Polarity Umpolung of Highly Electron-Rich P–H Bonds
M. B. Röthel, T. Eder, F. Brylak, M. Seidl, P. Löwe, F. Dielmann
Inorg. Chem. 2025; Link
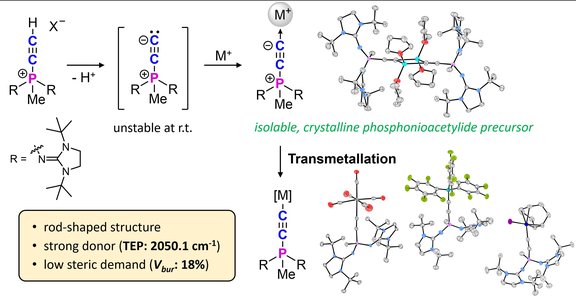
68. Accessing phosphonioacetylide chemistry: isolable alkali metal precursors for rod-shaped carbon donor complexes
F. Brylak, P. Löwe, K. Wurst, S. Hohloch, F. Dielmann
Inorg. Chem. Front. 2025, 12, 7556-7565; Advance Article; Link
67. Phosphonium Fullerides: Isolable Zwitterionic Adducts of a Phosphine with C60
M. B. Röthel, J. H. Franzen, D. Leitner, T. S. Hofer, M. Seidl, F. Dielmann
Chem. Sci. 2025, 16, 13189; Link

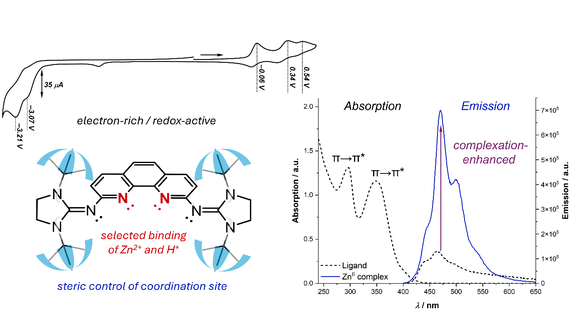
66. Electron-rich phenanthroline bearing N-heterocyclic imine substituents: synthesis, optical properties, metal coordination
J. H. Franzen, X. Zhou, K. Biv, A. Ajò, A. Mencke, L. FB Wilm, M. Seidl, T. Hofer, L. De Cola, P. Brüggeller, M. Thompson, F. Dielmann
Inorg. Chem. Front., 2025, 12, 6663-6675; Link
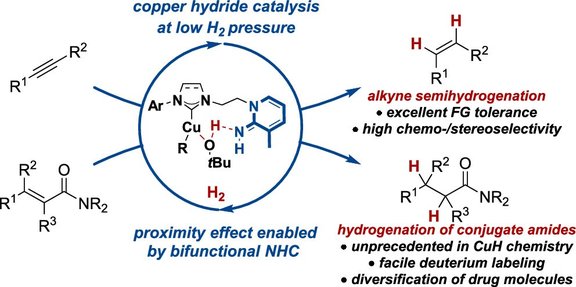
65. Broadly Applicable Copper(I)-Catalyzed Alkyne Semihydrogenation and Hydrogenation of α,β-Unsaturated Amides Enabled by Bifunctional Iminopyridine Ligands
M. Gorai, J. H. Franzen, P. Rotering, T. Rüffer, F. Dielmann, J. F. Teichert
J. Am. Chem. Soc. 2025, 147, 14481−14490, Link
63. Phosphonium SF5– Salts Derived from Sulfur Hexafluoride as Deoxyfluorination Reagents
M. B. Röthel, A. Schöler, F. Buß, P. Löwe, F. Dielmann
Chem. Eur. J. 2024, e202402028, Link

62. Light-Driven, Reversible Spatiotemporal Control of Dynamic Covalent Polymers
D. Reisinger, A. Sietmann, A. Das, S. Plutzar, R. Korotkov, E. Rossegger, M. Walluch, S. Holler-Stangl, T. S. Hofer, F. Dielmann, F. Glorius, S. Schlögl
Adv. Mater. 2024, 2411307, Link

61. Polyphosphorus Compounds Derived from P4 Conversion with Sterically Demanding Cobalt Complexes
F. Dielmann, M. E. Moussa, C. Riesinger, M. Scheer
z. Anorg. Allg. Chem. 2024, 650, e202400022, Link
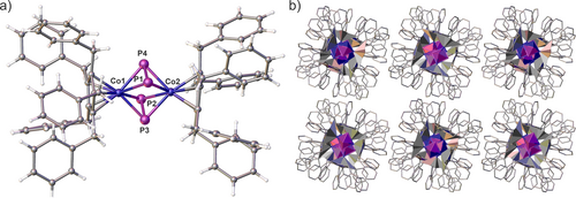
60. Electron-rich pyridines with para-N-heterocyclic imine substituents: ligand properties and coordination to CO2, SO2, BCl3 and PdII complexes
J. H. Franzen, L. F. B. Wilm, P. Rotering, K. Wurst, M. Seidl, F. Dielmann
Dalton Trans, 2024, 53, 11876-11883, Link

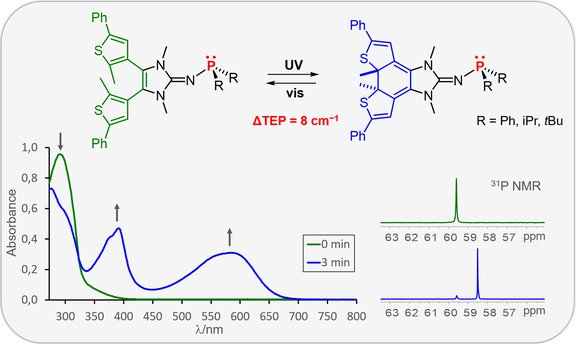
59. Photoswitchable Electron-rich Phosphines: Using Light to Modulate the Electron-Donating Ability of Phosphines
F. Buß, M. Das, D. Janssen-Müller, A. Sietmann, A. Das, L. F. B. Wilm, M. Freitag, M. Seidl, F. Glorius,* F. Dielmann*
Chem. Commun. 2023, 59, 12019–12022, Link
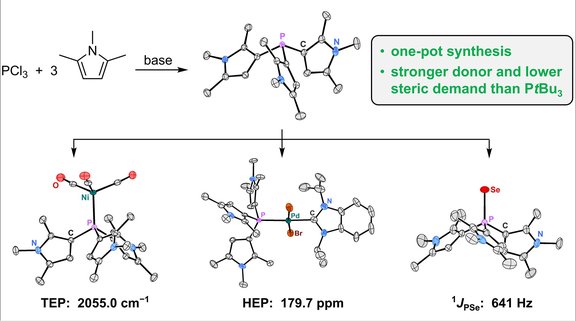
58. 1,2,5-Trimethylpyrrolyl Phosphines: A Class of Strongly Donating
Arylphosphines
J. A. Werra, K. Wurst, L. B. Wilm, P. Löwe, M. Röthel, F. Dielmann*
Organometallics 2023, 42, 597-605, Link
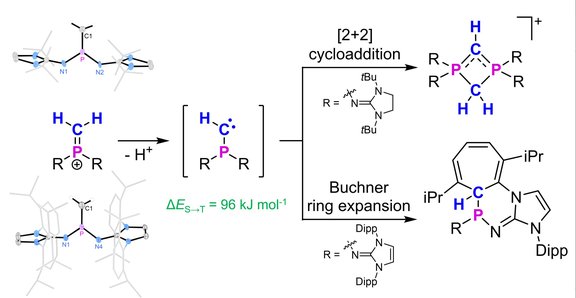
57. Terminal methylene phosphonium ions: precursors for transient
monosubstituted phosphinocarbenes
P. Löwe, M. A. Wünsche, F. R. S. Purtscher, J. Gamper, Th. S. Hofer, L. F. B. Wilm, M. Röthel, F. Dielmann*
Chem. Sci. 2023, DOI: 10.1039/D3SC02899B., Link
56. FacileOne-StepAccessto Pyrrole-Based1,4-Diphosphabarrelenes
J. Werra, K. Wurst, P. Löwe, F. Dielmann*
ChemPlusChem2023, 88, e2022004, Link
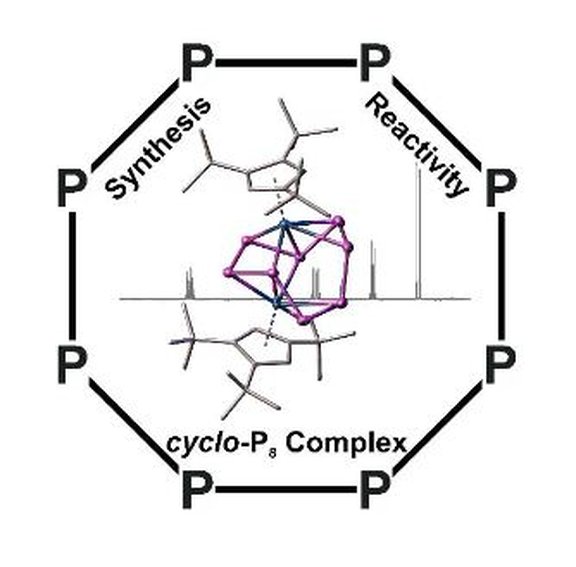
55. Synthesis and Reactivity of a Cyclo-Octatetraene-Like Polyphosphorus Ligand Complex [Cyclo-P8]
Ch. Riesinger, F. Dielmann, R. Szlosek, A. Virovets, M. Scheer*
Angew. Chem. Int. Ed.2023, e202218828, Link
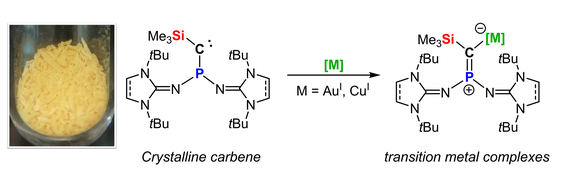
54. Crystalline phosphino(silyl)carbenes that readily form transition metal complexes
P. Löwe, F. Dielmann*
Chem. Commun. 2022, 58, 11831-11834; Link
53. Solvent-free photochemical decomposition of sulfur hexafluoride by phosphines: formation of difluorophosphoranes as versatile fluorination reagents
P. Rotering, C. Mück-Lichtenfeld, F. Dielmann*
Green Chem. 2022, 24, 8054-8061; Link

52. Reactivity of oxo- and thiophosphonium Lewis acids towards acetonitrile and pyridine
T.N. Parfeniuk, I.V. Kazakov, A.V. Pomogaeva, A.S. Lisovenko, P. Lӧwe, F. Dielmann, A.Y. Timoshkin*
Polyhedron 2022, 227, 116138; Link
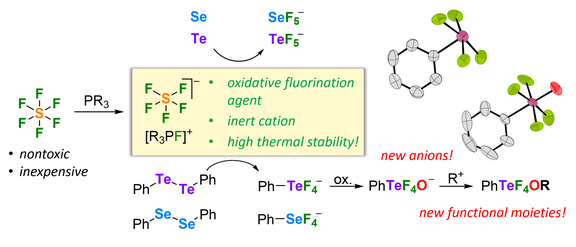
51. Oxidative Fluorination of Selenium and Tellurium Compounds using a Thermally Stable Phosphonium SF5- Salt Accessible from SF6
T. Eder, F. Buß, L. F. B. Wilm, M. Seidl, M. Podewitz, F. Dielmann*
Angew. Chem. Int. Ed. 2022, 61, e202209067; Link
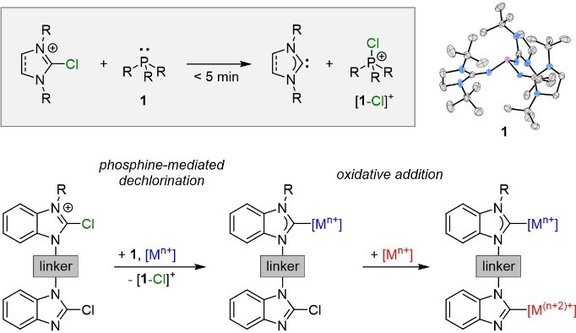
50. Synthesis of N-Heterocyclic Carbenes and Their Complexes by Chloronium Ion Abstraction from 2-Chloroazolium Salts Using Electron-Rich Phosphines
M. D. Böhme, T. Eder, M. B. Röthel, P. D. Dutschke, L. F. B. Wilm, F. E. Hahn,* F. Dielmann*
Angew. Chem. Int. Ed. 2022, 61, e20220219; Link
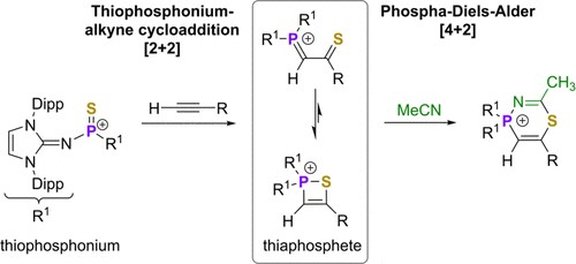
46. Thiophosphonium-Alkyne Cycloaddition Reactions: A Heavy Congener of the Carbonyl Alkyne Metathesis
P. Löwe, M. Feldt, M. B. Röthel, L. F. B. Wilm, F. Dielmann*
Inorg. Chem.2021, 60, 14509-14514; Link
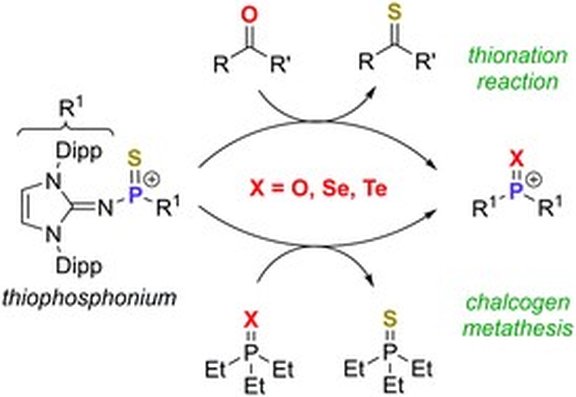
45. Lewis base-free thiophosphonium ion: a cationic sulfur atom transfer reagent
P. Löwe, T. Witteler, F. Dielmann*
Chem. Commun.2021, 57, 5043-5046; Link
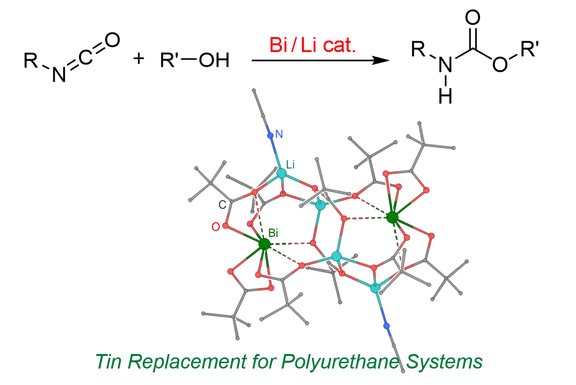
44. Heterobimetallic complexes composed of bismuth and lithium carboxylates as polyurethane catalysts – alternatives to organotin compounds
E. Levent, O. Sala, L. F.B. Wilm, P. Löwe, F. Dielmann*
Green Chem. 2021, 23, 2747-2755; Link
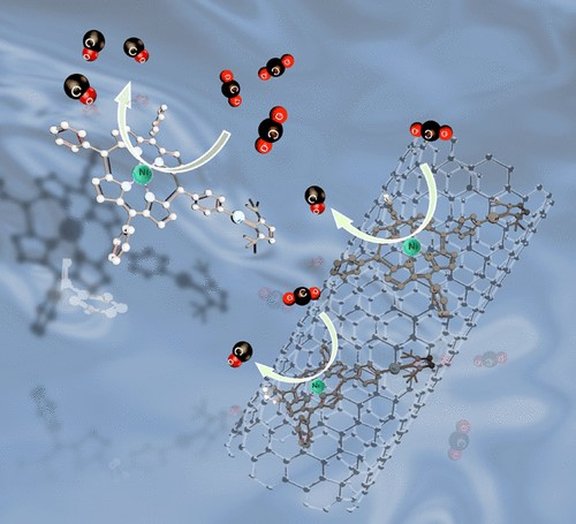
43. Electroreduction of CO2 catalyzed by nickel imidazolin-2-ylidenamino-porphyrins in both heterogeneous and homogeneous molecular systems
M. Abdinejad, L. F. B. Wilm, F. Dielmann,* H. B. Kraatz*
ACS Sustainable Chem. Eng. 2021, 9, 521-530; Link
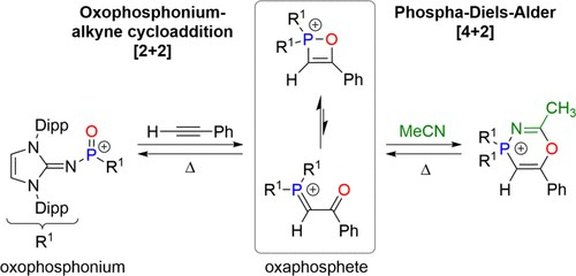
42. Oxophosphonium−Alkyne Cycloaddition Reactions: Reversible Formation of 1,2 Oxaphosphetes and Six-membered Phosphorus Heterocycles
P. Löwe, M. Feldt, M. A. Wünsche, L. F. B. Wilm, F. Dielmann*
J. Am. Chem. Soc. 2020, 142, 9818-9826; Link
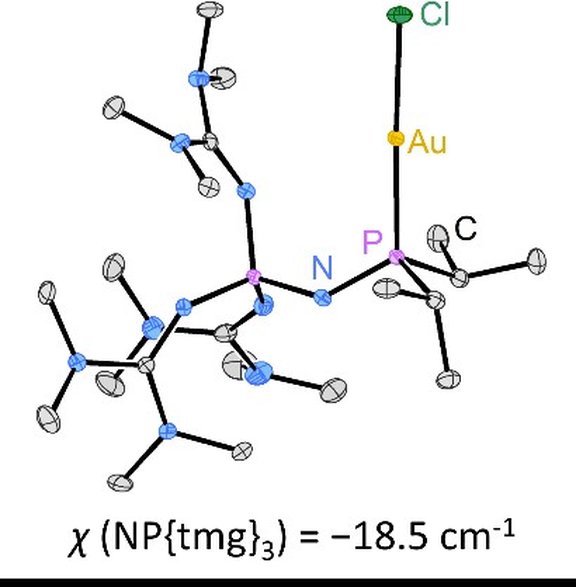
41. Effective control of the electron-donating ability of phosphines by using phosphazenyl and phosphoniumylidyl substituents
J. A. Werra, M. A. Wünsche, P. Rathmann, P. Mehlmann, P. Löwe, F. Dielmann*
Z. Anorg. Allg. Chem.2020, 646, 794-799; Link
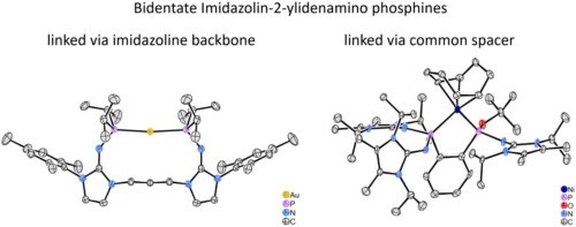
40. Synthesis and characterization of strongly electron-donating bidentate phosphines containing imidazolin-2-ylidenamino substituents and their electron-rich nickel(0), palladium(II) and gold(I) chelate complexes
L. F. B. Wilm, P. Mehlmann, F. Buß, F. Dielmann*
J. Organomet. Chem. 2020, 909, 1-8; Link
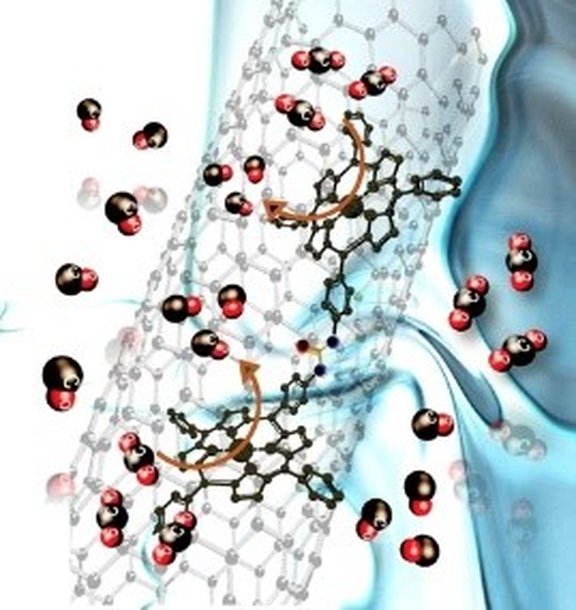
39. Enhanced Electrochemical Reduction of CO2 to CO upon Immobilization onto Carbon Nanotubes Using an Iron-Porphyrin Dimer
M. Abdinejad, C. Dao, B. Deng, M. E. Sweeney, F. Dielmann,* X. Zhang,* H. B. Kraatz*
ChemistrySelect 2020, 5, 979-984; Link
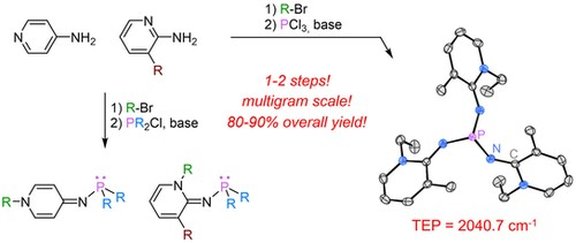
38. Pyridinylidenaminophosphines: Facile Access to Highly Electron-Rich Phosphines
P. Rotering, L. F. B. Wilm, J. A. Werra, F. Dielmann*
Chem. Eur. J.2020, 26, 406-411; Link
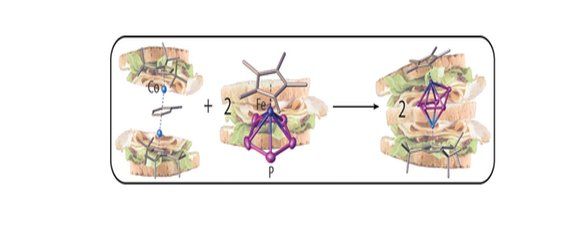
37. A general pathway to heterobimetallic triple-decker complexes
M. Piesch, F. Dielmann, S.Reichl, M. Scheer*
Chem. Eur. J. 2020, 26, 1518-1524; Link
36. Cooperative activation of azides by an Al/N-based active Lewis pair – unexpected insertion of nitrogen atoms into C–Si bonds and formation of AlCN3 heterocycles
W. Uhl,* K. Martinewski, J. Silissa Bruchhage, A. Hepp, M. Layh, F. Dielmann, P. Mehlmann
Z. Naturforsch.2020, 75b, 63-71; Link
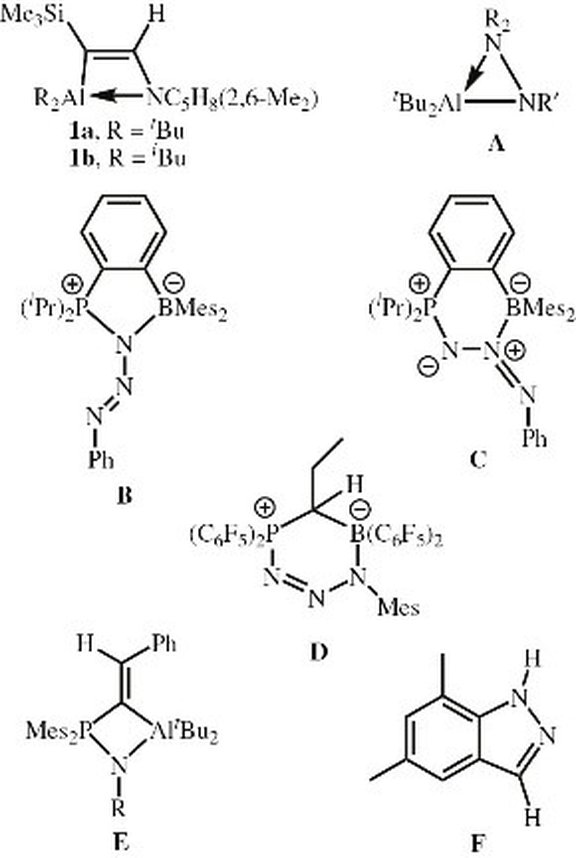
35. Isolation, characterization and reactivity of three-coordinate phosphorus dications isoelectronic to alanes and silylium cations
P.l Mehlmann, T. Witteler, L. F. B. Wilm, F. Dielmann*
Nat. Chem. 2019, 11, 1139-1143; Link
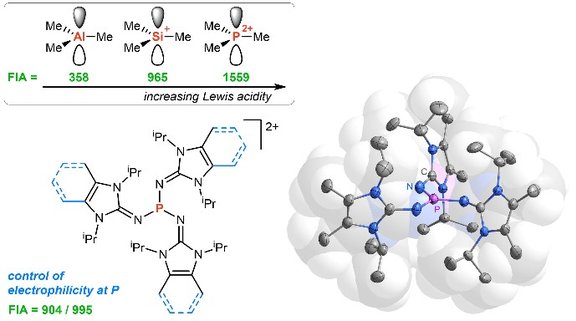
34. Homo- and Heterodinuclear Head-to-Head or Head-to-Tail Complexes of Rhodium(I) and Iridium(I) with C2,N3 or C8,N9 Bridging Azolato Ligands
S. Cepa, M. Böhmer, F. Roelfes, T. Tsai Yuan Tan, F. Dielmann, F. E. Hahn*
Organometallics 2019, 38, 1982-1990; Link
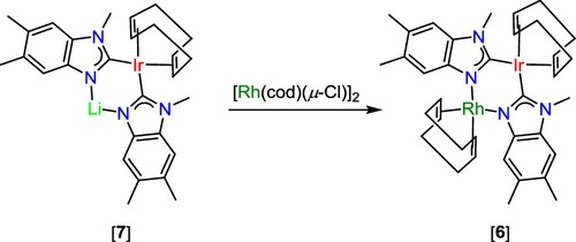
33. Reversible CO2 fixation by N-heterocyclic imines forming water-stable zwitterionic nitrogen-base–CO2 adducts
L. F. B. Wilm, T. Eder, C. Mück-Lichtenfeld, P.Mehlmann, M. Wünsche, F. Buß, F. Dielmann*
Green. Chem., 2019, 21, 640; Link
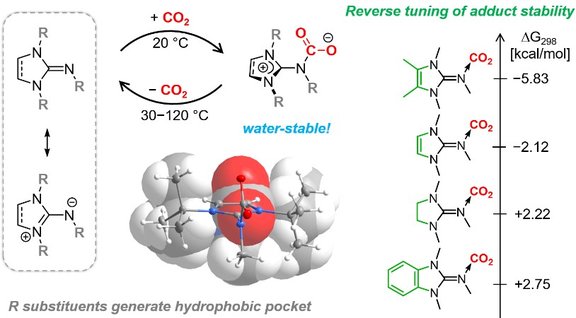
32. Switching the Electron-Donating Ability of Phosphines via Proton-Responsive Imidazolin-2 ylidenamino Substituents
P. Mehlmann, F. Dielmann*
Chem. Eur. J. 2019, 25, 2352-2357; Link
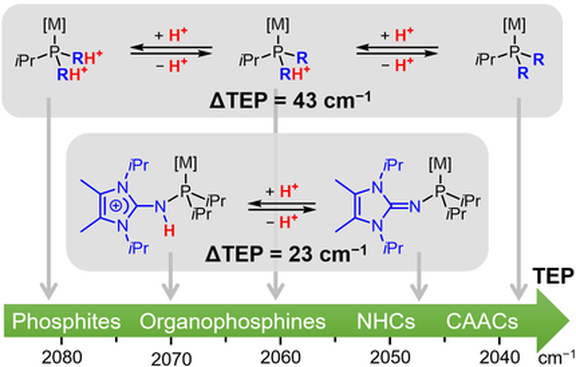
31. Dialkyl(1,3-diarylimidazolin-2-ylidenamino)phosphines: Strongly Electron-Donating, Buchwald-type Phosphines
T. Witteler, H. Darmandeh, P.Mehlmann, F. Dielmann*
Organometallics2018, 37, 3064-3072; Link
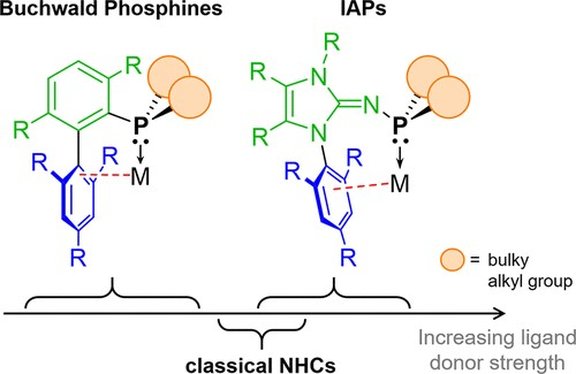
30. Crystalline, Room-Temperature Stable Phosphine‒SO2 Adducts: Generation of Sulfur Monoxide from Sulfur Dioxide
F. Buß, P. Rotering, Ch. Mück-Lichtenfeld, F. Dielmann*
Dalton Trans.2018, 47, 10420-10424; Link
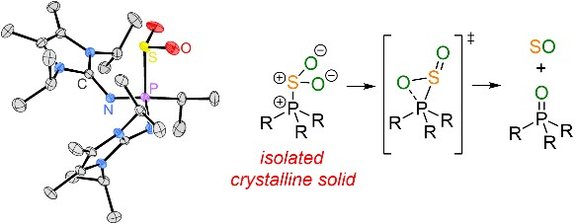
29. Lewis Base Free Oxophosphonium Ions: Tunable, Trigonal-planar Lewis Acids
M. Wünsche, T. Witteler, F. Dielmann*
Angew. Chem. Int. Ed. 2018, 57, 7234-7239; Angew. Chem. 2018, 130, 7354-7359; Link
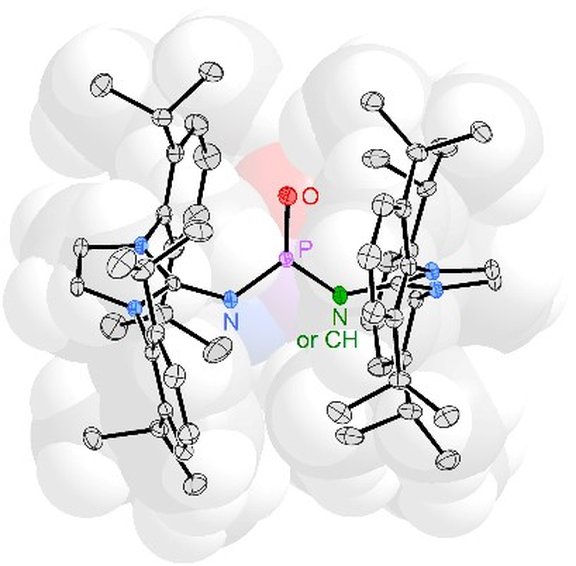
28. Nucleophilic Activation of Sulfur Hexafluoride: Metal-free, Selective Degradation by Phosphines
F. Buß, Ch. Mück-Lichtenfeld, P. Mehlmann, F. Dielmann*
Angew. Chem. Int. Ed. 2018, 57, 4951-4955; Angew. Chem.2018, 130, 5045-5049; Link
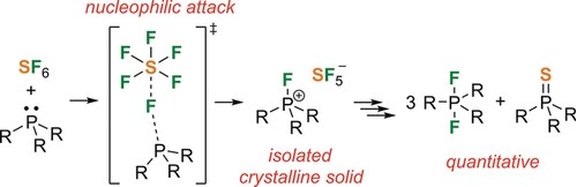
27. Stable Bromiranium Ions with Weakly-Coordinating Counterions as Efficient Electrophilic Brominating Agents
Ch. Ascheberg, J. Bock, F. Buß, Ch. Mück-Lichtenfeld, C. G. Daniliuc, K. Bergander, F. Dielmann, U. Hennecke*
Chem. Eur. J. 2017, 23, 11578–11586; Link
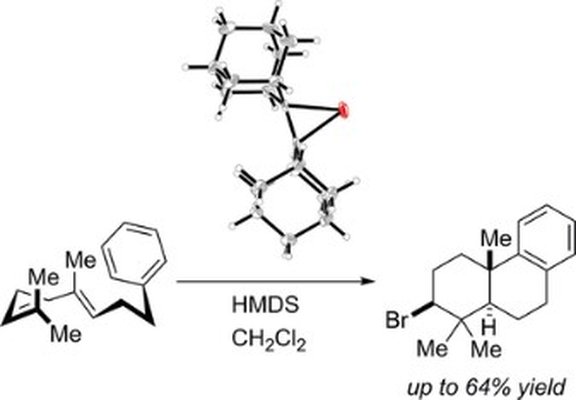
26. A tri-arylated 1,2,3-triazol-5-ylidene ligand with a redox-active ferrocenyl substituent for rhodium(I) catalyzed hydroformylation of 1-octene
D. Aucamp, T. Witteler, F. Dielmann, S. Siangwata, D. C. Liles, G. S. Smith, D. I. Bezuidenhout*
Eur. J. Inorg. Chem. 2017, 1227-1236; Link
25. Quantifying the Efficiency of CO2 Capture by Lewis Pairs
J. J. Chi, T. C. Johnstone, D. Voicu, P. Mehlmann, F. Dielmann, E. Kumacheva,* D. W. Stephan*
Chem. Sci.2017, 8, 3270-3275; Link

24. Tris(imidazolin-2-ylidenamino)phosphine: A crystalline phosphorus(III) superbase that splits carbon dioxide
P. Mehlmann, Ch. Mück-Lichtenfeld, T. T. Y. Tan, F. Dielmann*
Chem. Eur. J. 2017, 23, 5929-5933; Link
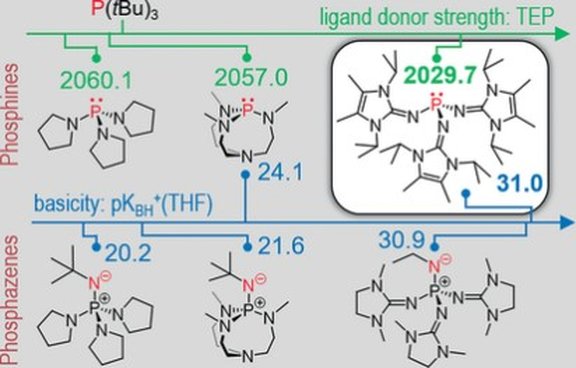
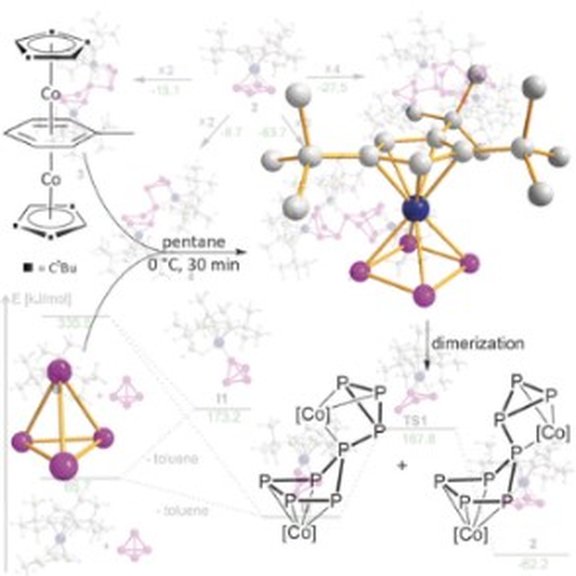
23. The Cobalt cyclo-P4 Sandwich Complex and Its Role in the Formation of Polyphosphorus Compounds
F. Dielmann, A. Timoshkin, M. Piesch, G. Balázs, M. Scheer*
Angew. Chem. Int. Ed. 2017, 56, 1671-1675; Angew. Chem. 2017, 129, 1693-1698; Link
22. Reversible Carbon Dioxide Binding by Simple Lewis Base Adducts with Electron-Rich Phosphines
F. Buß, P. Mehlmann, Ch. Mück-Lichtenfeld, K. Bergander, F. Dielmann*
J. Am. Chem. Soc.2016, 138, 1840-1843; Link
21. Tetrahedrally coordinated luminescent copper(I) compounds containing halide, phosphane and norharmane ligands
R. Ahmad Khan, F. Dielmann, X. Liu, F. E. Hahn, K. Al-Farhan, A. Alsalme, J. Reedijk*
Polyhedron 2016, 111, 173-178; Link
20. Synthesis, structure and spectroscopic properties of bis(triphenylphosphane)iminium (chlorido)(cyanido)argentates(I)
M. Jaafar, X. Liu, F. Dielmann, F. E.Hahn, K. Al-Farhan, A. Alsalme and J. Reedijk*
Inorg. Chim. Acta 2016, 443, 45-50; Link
19. Cyclo-P4 Building Blocks: Achieving Non-Classical Fullerene Topology and Beyond
F. Dielmann, E. V. Peresypkina, B. Krämer, F. Hastreiter, B. P. Johnson, M. Zabel, C. Heindl, and M. Scheer*
Angew. Chem. Int. Ed. 2016, 55, 14833-14837; Angew. Chem. 2016, 128, 15053-15058; Link
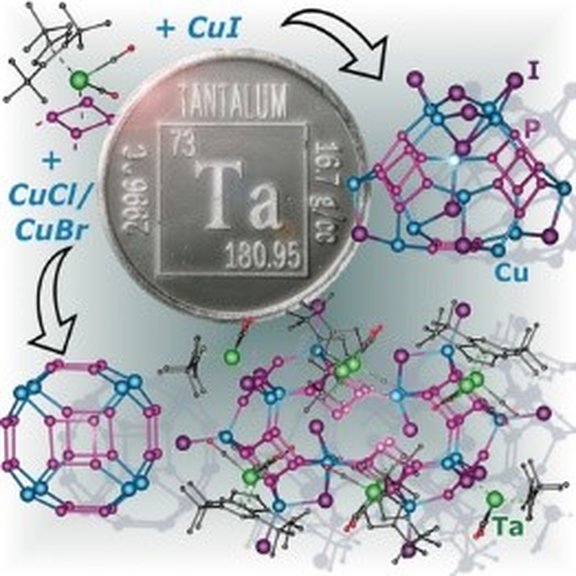
18. Oxidation Chemistry of Inorganic Benzene Complexes
M. Fleischmann, F. Dielmann, G. Balázs and M. Scheer*
Chem. Eur. J. 2016, 22, 15248-15251; Link
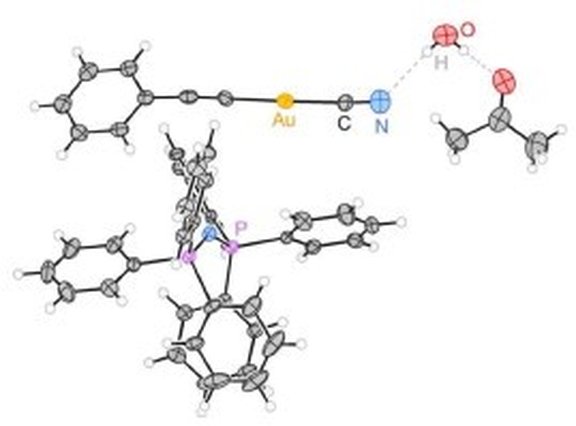
17. Synthesis, structure and spectroscopic properties of bis(triphenylphosphane)iminium(phenylacetylido)(cyanido)aurate(I) monoacetone monohydrate, (PPN)[Au(CN)(CCC6H5)]·H2O·(CH3)2CO and bis(triphenylphosphane)iminium (t-butylacetylido)(cyanido)aurate(I) monohydrate, (PPN)[Au(CN)(CCC4H9)]·H2O
A. Alsalme, M. Jaafar, X. Liu, F. Dielmann, F. E. Hahn, K. Al-farhan, J. Reedijk*
Polyhedron 2015, 88, 1-5; Link
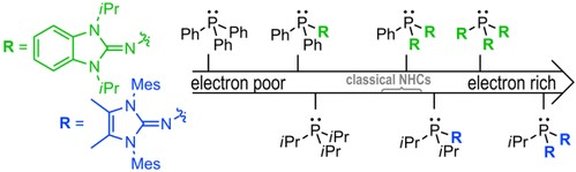
16. Imidazolin-2-ylidenaminophosphines as Highly Electron-Rich Ligands for Transition-Metal Catalysts
M. A. Wünsche, P. Mehlmann, T. Witteler, F. Buß, P. Rathmann, F. Dielmann*
Angew. Chem. Int. Ed. 2015, 54, 11857-11860; Angew. Chem. 2015, 127, 12024-12027; Link
15. Reactivity of a Stable Phosphinonitrene towards Small Molecules
F. Dielmann and G.Bertrand*
Chem. Eur. J. 2015, 21, 191−198; Link
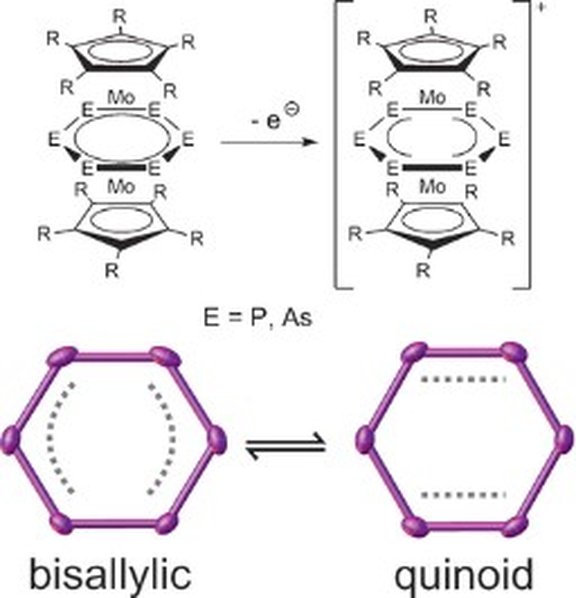
14. Redox and Coordination Behavior of the Hexaphosphabenzene Ligand in [(Cp*Mo)2(,6:6-P6)] Towards the “Naked” Cations Cu+, Ag+, and Tl+
M. Fleischmann, F. Dielmann, L. J. Gregoriades, E. V. Peresypkina, A. V. Virovets, S. Huber, A. Y. Timoshkin, G. Balázs, and M. Scheer*
Angew. Chem. Int. Ed. 2015, 54, 13110-13115; Angew. Chem.2015, 127, 13303-13308; Link
13. Tunable Porosities and Shapes of Fullerene-Like Spheres
F. Dielmann, M. Fleischmann, C. Heindl, E. V. Peresypkina, A. V. Virovets, R.M. Gschwind, and M. Scheer*
Chem. Eur. J. 2015, 21, 6208−6214; Link
12. Isolation of Bridging and Terminal Coinage Metal-Nitrene Complexes
F. Dielmann, D. M. Andrada, G. Frenking, G. Bertrand*
J. Am. Chem. Soc.2014, 136, 3800−3802; Link
11. A Nano-sized Supramolecule Beyond the Fullerene Topology
F. Dielmann, C. Heindl, F. Hastreiter, E. V. Peresypkina, A. V. Virovets, R. M. Gschwind, and M. Scheer*
Angew. Chem. Int. Ed. 2014, 53, 13605−13608; Angew. Chem. 2014, 126, 13823−13827; Link
10. Crystalline, Lewis Base-Free, Cationic Phosphoranimines (Iminophosphonium Salts)
F. Dielmann, C. E. Moore, A. L. Rheingold, G. Bertrand*
J. Am. Chem. Soc. 2013, 135, 14071−14073; Link
9. Intramolecular Phosphorus-Phosphorus Bond Formation within a Co2P4 Core
T. Li, N. Arleth, M. T. Gamer, R. Köppe, T. Augenstein, F. Dielmann, M. Scheer, S.N. Konchenko, P. W. Roesky*
Inorg. Chem. 2013, 52, 14231−14236; Link
8. A Crystalline Singlet Phosphinonitrene: A Nitrogen Atom–Transfer Agent
F. Dielmann, O. Back, M. Henry-Ellinger, P. Jerabek, G. Frenking, G. Bertrand*
Science 2012, 337, 1526−1528; Link
7. Coordination Polymers Based on [Cp*Fe(η5-P5)]: Solid-State Structure and MAS NMR Studies
F. Dielmann, A. Schindler, S. Scheuermayer, J. Bai, R. Merkle, M. Zabel, A. V. Virovets, E. V. Peresypkina, G. Brunklaus, H. Eckert, M. Scheer*
Chem. Eur. J. 2012, 18, 1168−1179; Link
6. Access to Extended Polyphosphorus Frameworks
F. Dielmann, M. Sierka, A. V. Virovets, M. Scheer*
Angew. Chem., Int. Ed. 2010, 49, 6860−6864; Angew. Chem.2010, 122, 7012−7016; Link
5. Synthesis and reactivity of low-valent group 14 element compounds
B. P. Johnson, S. Almstätter, F. Dielmann, M. Bodensteiner, M. Scheer*
Z. Anorg. Allg. Chem. 2010, 636, 1275−1285; Link
4. New pentaphosphaferrocenes
F. Dielmann, R. Merkle, S. Heinl, M. Scheer*
Z. Naturforsch. 2009, 64b, 3−10; Link
3. Dimolybdenum Bis-2,4,6-triisopropyl-benzoate Bis-4-isonicotinate: A Redox Active Analogue of 4,4′-Bipyridine with Ambivalent Properties
M. H. Chisholm, A. S. Dann, F. Dielmann, J. C. Gallucci, N. J. Patmore, R. Ramnauth, M. Scheer*
Inorg. Chem. 2008, 47, 9248−9255; Link
2. Dynamic Investigation on Supramolecular Compounds using Pn ligand complexes
M. Scheer,* L. J. Gregoriades, R.Merkle, B. P. Johnson, F. Dielmann
Phosphorus, Sulfur and Silicon and the Related Elements2008, 183, 504−508; Link
1. Spherical Cluster Comprising a Four- and Six-Membered-Ring Motif
B. P. Johnson, F. Dielmann, G. Balázs, M. Sierka, M.Scheer*
Angew. Chem. Int. Ed. 2006, 45, 2473−2475; Angew. Chem.2006, 118, 2533−2536; Link
Patents
9. Methods for decomposing sulfur hexafluoride
F. Dielmann, P. Rotering
European patent application (Nr. EP 21 194 663.7, application date 02.09.2021).
8. Electron-rich carboxylates as ligands for Lewis acidic metal complexes as catalysts in the polyurethane reaction
F. Dielmann, P. Hoffmann, E. Levent
European patent application (Nr. EP 20 189 240.3, application date 03.08.2020)
7. Zinkhaltiger Katalysator umfassend mindestens einen aromatischen Substituenten
F. Dielmann, P. Hoffmann, E. Levent
European patent application (Nr. EP 20 189 240.3, application date 03.08.2020).
PCT Int. Appl. (2022), WO 2022028850 A1 20220210
6. Mixtures comprising bismuth-containing catalysts and diols
F. Dielmann, P. Hoffmann, E. Levent
European patent application (Nr. EP 20 153 336.1, application date 23.01.2020).
PCT Int. Appl. (2021), WO2021148332 A1 20210729.
5. Process to the fluorination of organoelement compounds
F. Buß, F. Dielmann, M. Wünsche
German patent application (Nr. 10 2019 108 416.4, application date 01.04.2019).
Ger. Offen. (2020), DE 102019108416 A1 20201001.
4. Bismuth-containing catalyst comprising at least one aromatic substituent
F. Dielmann, P. Hoffmann, E. Levent
European patent application (Nr. 19155916.0, application date: 07.02.2019).
PCT Int. Appl. (2020), WO 2020160939 A1 20200813.
3. Air cooler
F. Buß, F. Dielmann, M. Wünsche
German patent application (Nr. 10 2017 124 711.4, application date: 23.10.2017).
Ger. Offen. (2019), DE 102017124711 A1 20190425.
2. Methods for the degradation of SF6
F. Buß, F. Dielmann
International patent (No: WO/2019/077086, International application number PCT/EP2018/078652, application date: 19.10.2017).
PCT Int. Appl. (2019), WO 2019077086 A1 20190425.
1. Gasdichter Gewindeansatz und Verschlussstempel für NMR-Rohre
J. Berendes, F. Buß, F. Dielmann, F. Henrichmann, M. Wünsche
German utility model (Nr. 20 2017 004 691, application date: 08.09.2017, Entry date: 15.12.2017).
Editorials
PhD Thesis
Novel phosphorus rich complexes and their use in supramolecular chemistry
Fabian Dielmann (Dissertation)
München: Verlag Dr. Hut, 2011 [ISBN 9783868538946].