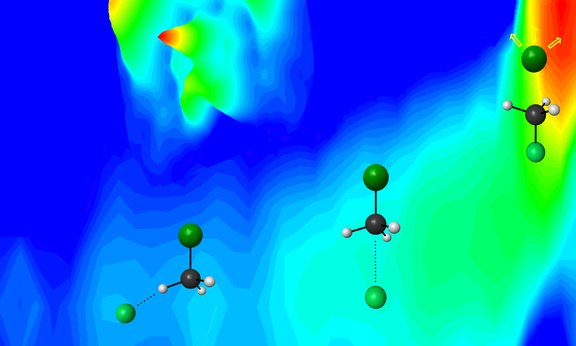
One of the most studied reactions in physical organic chemistry is the nucleophilic substitution reaction. This process plays an important role in organic synthesis such as in the production of vitamins or the formation of adrenaline in living cells. In the course of the reaction, an atom or a group of atoms bound to a molecule is exchanged by another binding partner. The attacking reaction partner, the nucleophile, possesses electrons that are ready to start a reaction. They allow it to form a new bond to a central carbon atom. This starts a substitution reaction and in its course the former partner is repelled and the molecule forms a new bond with the attacker.
For many decades chemists and physicists have sought to understand these processes on a fundamental atomic level and study them in laboratory experiments. Now Roland Wester and his research group at the Institute for Ion Physics and Applied Physics of the University of Innsbruck, Austria have managed to discover surprising new aspects in the dynamics of this reaction, which were previously considered to be well understood.
Reaction dynamics
In cooperation with the theory group of Gabor Czako at the University of Szeged, Hungary, the combination of experiment and simulation allowed the unravelling of the subtle role played by the repelled partner, the leaving group, during the course of a substitution reaction. For this the researchers studied the substitution of negatively charged halogen atoms in the most basic organic molecular structure, the methyl group CH3.
Wester and colleagues used two crossed beams that bring the neutral reactant molecules and the attacking halogen ions to collision. The ions that are repelled during the reaction were analysed with detectors that record the velocity of each reaction product. Jennifer Meyer, postdoctoral researcher in the Wester group and one of the co-authors of this article stresses the surprising observations: „In conjunction with the simulations for this reaction we found strong differences between the dynamics observed for different repelled partners.“ From this it could be deduced how much the halogen ion is able to influence the neutral molecule prior to the substitution, either by orienting it or by exciting internal vibrational modes. Overall, the initial partner influences, in an unexpectedly marked way, if and how the attacking ion can break up an established bond.
Links
- Influence of the leaving group on the dynamics of a gas phase SN2 reaction. Martin Stei, Eduardo Carrascosa, Martin A. Kainz, Aditya H. Kelkar, Jennifer Meyer, István Szabó, Gábor Czakó and Roland Wester. Nature Chemistry, Advance Online Publication 2015
- Molecular Systems group, Institute for Ion Physics und Applied Physics