Nanostructured Model Catalysis
Our group focuses on the mechanistic understanding of processes at the solid/gas interface on materials relevant for reactions in sustainable catalysis, such as methanol steam or methane dry reforming and the selective catalytic reduction of nitrogen oxide. Materials range from oxide over metal-oxide systems to intermetallic compounds and alloys and by combining model systems under ultra-high vacuum and (powder) materials under technologically relevant conditions we are devoted to close the “pressure” and “materials” gap in catalysis. Our approach is the exclusive use of in situ and operando structural and spectroscopic techniques to study the catalyst behavior under close-to-real conditions.
This interdisciplinary approach involves collaboration between researchers from various fields, including materials science, chemistry, physics, and engineering. We strive to develop innovative solutions to complex scientific problems and contribute to the advancement of the field.
News
We are pleased to announce that, due to the successful implementation of two new projects, we have open PhD positions available. We are seeking motivated and talented young scientists who are interested in pursuing a PhD in one of our projects.
The two projects offer exciting research opportunities in various fields and enable PhD students to work on innovative topics and gain valuable experiences. We provide a challenging and stimulating research environment as well as intensive supervision by experienced scientists.
If you are interested in pursuing a PhD in one of our projects, we look forward to receiving your application. Please check the announcements on our website and submit your application documents by the specified deadline. We are excited to welcome talented and dedicated young researchers to our team.
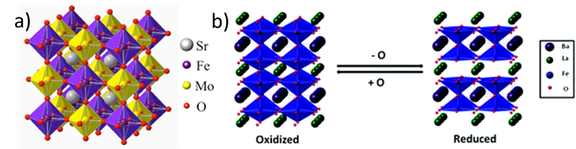
Double Perovskites as Mixed Ionic-Electronic Conductors FWF P 35770
The main focus of the project “Double Perovskite-based Mixed Ionic-Electronic Conductors (MIEC)” lies on the synthesis and understanding of a new prospective class of double perovskite systems on strontium-iron-vanadium/niobium basis (Sr2Fe1+x(V/Nb)1-xO6-d) with controlled defect chemistry, and their in situ and operando characterization with respect to their use as prospective MIEC materials in solid oxide cells. Double perovskites belong to the class of complex oxides, which - due to their manifold physico-chemical properties – exhibit the potential to substitute to date used conventional materials with unfavorable behavior under extreme experimental conditions in fuel cell systems. To do so, the usual main obstacle of poor structural stability under relevant technological operating conditions has to be overcome. The focus of the project is the direct correlation of in situ/operando-determined redox chemistry and structural stability with catalytic properties in internal reforming reactions. The most important step hereby is the knowledge-based synthesis of the materials under controlled oxygen fugacity to directly steer the defect concentration and the redox behavior. In this respect, we will directly exploit the physico-chemical properties of the constituting elements to influence and trigger favorable material’s properties. Of equal and paramount importance is the structural and (electro-) chemical characterization of the materials under in situ and operando conditions, i.e. in the state of operation.
To achieve these goals, we utilize a portfolio of characterization methods capable of being operated under such close-to-real technological conditions. This includes in situ/operando structural characterization (X-ray diffraction, electron microscopy), electrochemical performance (electrochemical impedance spectroscopy), surface chemical characterization (FT-Infrared Spectroscopy), monitoring the electronic structure (in situ X-ray photoelectron spectroscopy) and quantitatively assessing the defect concentration. The scientific and methodological approach allows us to gain unprecedented insight into the structure-property relationships of double-perovskite materials in the working state of the materials and to optimize generalized synthesis routines to prospective new catalytic materials for SOC operation.
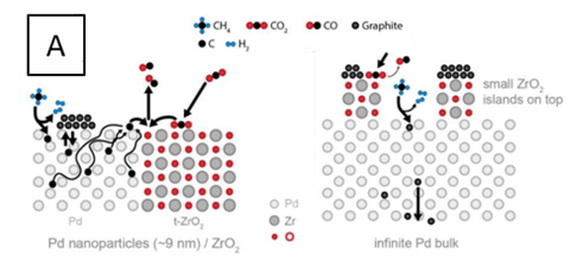
Optimization of Carbon Chemistry in Methane Dry Reforming FWF P 36926-N
The main goal of the project “Optimization of the Carbon Chemistry in Methane Dry Reforming” is connected to the in situ/ operando experimental assessment and fundamental understanding of reactive carbon intermediates in different branches of the methane dry reforming network. In depth understanding of carbon reactivity and coke suppression will enable a focused, knowledge-based design of methane dry reforming. The central focus of the project is the direct correlation of the in situ/ operando determined reactivity of different carbon species with catalytic methane dry reforming activity. By exploiting the controlled in situ decomposition of selected intermetallic compounds towards active metal-oxide interfaces, we are able to quantify the reactivity of carbon bound in variable oxidation states between carbides and oxy-carbonates, including the suppression and re-mobilization of coke. The influence of material-specific redox chemistry, involving the the elementary carbon-forming and -converting reactions within the dry reforming network, will be scrutinized. To extract detailed structure-reactivity relationships, we will complement kinetic reactivity studies with in situ/ operando bulk and surface characterization under realistic reaction conditions. To accomplish these tasks, we rely on an exclusive portfolio of in situ and operando characterization methods capable of operation under close-to-technological conditions. This includes in situ/operando structural (X-ray diffraction, electron microscopy, Raman spectroscopy) and surface chemical characterization (X-ray photoelectron spectroscopy and FT-Infrared Spectroscopy), as well as integral quantification of carbon (thermo-gravimetry). We complement this characterization with kinetic reactor studies of important reactions within the dry reforming network and with theoretical assessment of the observed carbon reactivity trends. The controlled in situ decomposition of variable intermetallic precursors allows to induce a broad range of interfacial and bulk carbon species. Our methodological approach will provide novel material-specific insights into viable carbon reaction pathways within the methane dry reforming network.
Contact
Institut für Physikalische Chemie
Josef-Möller-Haus
Innrain 52c
A - 6020 Innsbruck
Österreich
Teamleaders
Assoz. Prof. Dr.
Bernhard Klötzer
Priv.Doz- Dr.
Simon Penner

