Subcellular LOcalization of Toxin production in cyanobacteria (SLOT)
Funded by the Austrian Science Fund (FWF)
https://www.fwf.ac.at/forschungsradar/10.55776/P32193
October 2019 – March 2024
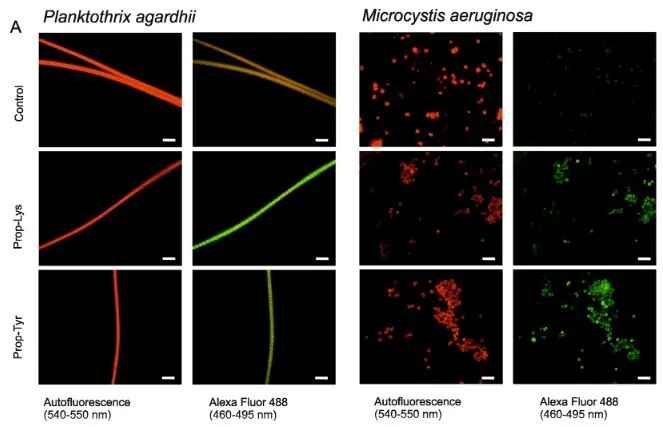
Microphotographs of cyanobacteria Planktothrix agardhii and Microcystis aeruginosa recorded under two emission spectra (autofluorescence and Alexa Fluor 488) using epifluorescence microscopy (100-fold magnification). Bar is 10 µm.
Control cells (cells grown and processed under identical conditions but without substrate enabling so called biorthogonal labeling); Prop-Lys, Prop-Tyr, cells fed with substrates enabling bioorthogonal labeling. From Kurmayer et al. (2020) Scientific Reports https://doi.org/10.1038/s41598-020-59381-w
Introduction
Cyanobacteria are found in many different aquatic or terrestrial environments such as lakes, oceans and rivers, soil and cliffs or more extreme habitats such as geysers or biologicals crusts formed in arctic or aride climate. The ability to form blooms in lakes and rivers under certain circumstances (such as water stratification and eutrophication) and the rich metabolic pathways to produce toxins (microcystins, nodularin, and others) and bioactive metabolites (anabaenopeptides, aeruginosins, cyanopeptolins, among others) make them an interesting subject of study.
In general the changing climate is leading us to conditions where cyanobacterial blooms are going to be more prolific , affecting the usage of water sources and recreation. Certain cyanobacteria such as Microcystis and Planktothrix form algal blooms associated with potentially high concentrations of the above mentioned toxins causing human diseases and death of animals either through drinking water exposure or the food chain. Thus those blooms are nowadays called cyanobacterial harmful algal blooms (cHAB) because containing cyanotoxins.

Landsat 8 real color image of cHAB formed by Nodularia spumigena in the Baltic Sea (23-July-2018)
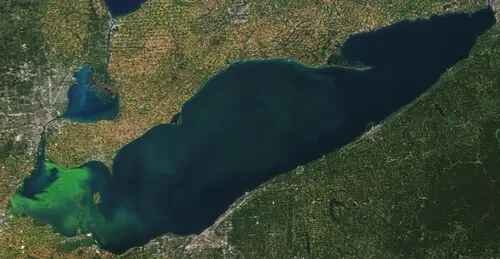
Lake Erie Bloom in Michigan, Ohio (24-Sept-2017) formed by Microcystis aeruginosa and Planktothrix agardhii. NASA Earth observatory
The most common toxins produced by cyanobacteria are the microcystins, with more than 200 described structural variants. Microcystins are hepatotoxic heptapeptides produced by non-ribosomal peptide synthetases (NRPS), using (non-)proteinogenic amino acids. The microcystin general structure is composed of (1)D-Ala-(2)X-(3)D-MeAsp-(4)Z-(5)Adda-(6)D-Glu-(7)Mdha, where X and Z denote variable L-amino acids. Microcystins and nodularin are toxic to eukaryotic organisms because of the inhibition of protein phosphatases 1 and 2A. Various (not necessarily) exclusive functions have been proposed, reaching from protection against predators and chemical defense to the protection of cellular proteins under oxidative stress conditions.
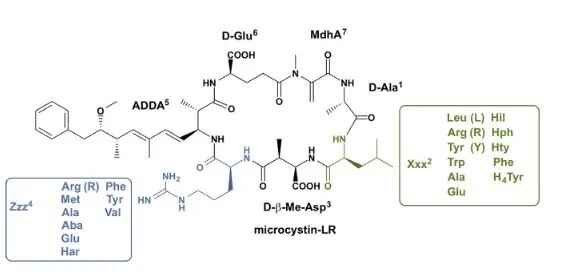
Microcystin molecule conserved structure and its variation. Meyer et al. (2016), Cell Chemical Biology, DOI: https://doi.org/10.1016/j.chembiol.2016.03.011
Analogous to microcystins the anabaenopeptides are bioactive hexapeptides produced by NRPS and are comprised of a ring of five amino acids fused to an exocyclic amino acid through the ureido linkage at the conserved D-Lys. As a group the anabaenopeptides show variability in bioactivity, such as acting as inhibitors of carboxypeptidases A and B, protein phosphatase 1, and serine proteases such as (chymo)trypsin.
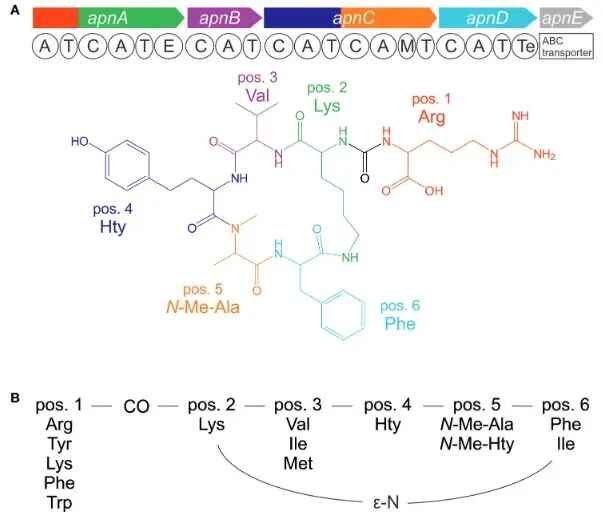
Anabaenopeptide molecule general structure and the genetic basis of its synthesis. Entfellner et al. (2017) Front. Microbiol. https://doi.org/10.3389/fmicb.2017.00219
These peptides both are found widely spread among aquatic cyanobacteria occurring in freshwater and marine habitats as well as lower invertebrates such as sponges. Among terrestrial cyanobacteria prominent representatives are found in lichens and soil-dwelling cyanobacteria. Understanding the regulation of toxin or bioactive peptide synthesis at the (sub)cellular level of various genotypes is considered essential to understand the factors leading to the genetic variability observed in blooms, such as the co-occurrence of toxic and nontoxic genotypes in water (e.g. Chen et al. 2019, BMC Microbiology, https://doi.org/10.1186/s12866-016-0639-1). In general these peptides occur at high intracellular concentrations (i.e. one promille to half percent of cellular dry weight) and are potentially actively or passively released into the water. The conditions when those peptides are transported out of the cell are not known but would be also relevant to protection and water management.
Aims
Subcellular location of microcystins and anabaenopeptides.
It has been suggested that the Adda moiety of the microcystins binds to membranes because of the lipohilic nature. In fact, a structural variety of microcystins, nodularin has been reported to form pores in lipid bilayers and native membranes. Further, using immune-gold labeling techniques the majority of the microcystins have been detected surrounding the thylakoid area. Furthermore proteomic assays have confirmed covalent linkage between microcystins and abundant proteins such a phycobiliproteins and Rubisco. Since thylakoid structures can be visualized through autofluorescence and distinguished from chemically labeled peptides we can investigate the location of the peptides more directly using advanced imaging. Using higher microscopic resolution will further help to find possible in vivo compartmentation, or if the peptides are located in the cytoplasm or the periplasmic space.
Intercellular variation of microcystin/anabaenopeptin synthesis.
It has been shown that the gene cluster of microcystin synthesis is prone to transposase driven mutations (insertions or deletions), thus altering the microcystin synthesis at a cellular level. Analogously mutations have been found for the gene cluster encoding anabaenopeptin synthesis. The chemical labelling techniques can be a prominent tool to detect and quantify the variability of the different peptide signal among the cells and genotypes.
In vivo observation of intracellular microcystin and anabaenopeptide pool and its potential release.
Previously the intra and extracellular distribution of peptides has been studied using strains, revealing significant difference between the intra and extracellular content of peptides. It is speculated that the strains with higher extracellular peptide content are actively exporting or show other specific difference in cell wall permeability. We will investigate active or passive transport processes by imaging chemically labeled peptides using microscopy, flow-cytometry, and LC-MS quantification.
In vivo observation of the fate of intracellular peptides during physiological stress conditions.
It is known that the physiological stress affects the allelopathy in many different organisms. Using chemical labeling we aim to differentiate the effects of contrasting physiological growth conditions of the intracellular peptides and their release.
News
December 2024:
Kristin Kuntz, MSc Molecular Biology, Univ. of Innsbruck has successfully submitted her MSc thesis
“Subzelluläre Lokalisierung der Toxinproduktion in Cyanobakterien: Eine umfassende Analyse der Signalintensität und Signalverteilung in der Zelle bei Microcystis aeruginosa und Planktothrix agardhii“, (107 pp).
and will defend on 20 December 2024.
May 2024:
Welcome to our Long Night of Research (Lange Nacht der Forschung), 24 May 2024 in Mondsee
Station: Are you interested to observe the production of toxins in the cells of blue-green algae? Many blue-green algae (cyanobacteria) with toxic ingredients are known to be present in water bodies. These poisons, especially microcystins and anabaenopeptins, can also have negative effects on higher organisms in larger quantities.
We specifically use click chemistry technology to observe toxins in individual cells. This makes it possible to detect this production via bright dyes in the cells. Experience this first-ever application of click chemistry to observe poison peptides directly in the cells!
March 2024:
The project finished at 31 March 2024. The Corona-related extension of the project duration by the 18 months was necessary in order to make up for the lost laboratory work due to Corona measures.
January 2024:
Welcome to Loay Hussein who will join the project team through a 5 months internship to fulfill the Master 2 Graduate program in Evolutionary Biology at the University of Lille in France.
June 2023:
Kristin Kuntz started her MSc thesis on “Subcellular localization of toxin production in cyanobacteria through compartmentalization analysis”. Welcome on board and good luck!
Poster-Presentation at Gordon Research Conference on Mycotoxins and Phycotoxins 2023, Easton, 19.06.2023: Moron-Asensio, R. & Kurmayer, R.: Monitoring intracellular synthesis and release of microcystins (MC) in bloom forming cyanobacteria Microcystis aeruginosa.
April 2023:
Our project technician David Schuler (TA position, since December 1st, 2019) resigned from his function on 1st May. Fortunately on 1st April Kathrin Geir took over as a student coworker. We are grateful for this flexibility!
December 2022:
Daniel Holzer started his MSc thesis on “Real-time observation of mutations by transposases in toxin synthesis genes in cyanobacteria”. Welcome and good luck!
November 2022:
MSc thesis Christoph Kotzorek has been approved:
Kotzorek, C. (2022). Click labelling and In Vitro clicking (CuAAC) of the widespread cyanotoxin Microcystin and the bioactive peptide Anabaenopeptin from cyanobacteria, MSc thesis University of Innsbruck, pp. 95 (supervision: R. Kurmayer)
September 2022, presentation:
Morón-Asensio, R. co-authors: Kurmayer, R.: Localization and duration of the synthesis of chemically-modified microcystins/bioactive peptides in the bloom-forming cyanobacteria Microcystis aeruginosa and Planktothrix agardhii. 7th early career research symposium on cyanobacteria (Cyano2022), Leipzig, 27.09.2022.
August 2022, presentation:
Morón-Asensio, R. co-authors: Kurmayer, R.: Localization and duration of the synthesis of chemically-modified microcystins/bioactive peptides in the bloom-forming cyanobacteria Microcystis aeruginosa and Planktothrix agardhii. 36th Congress of the International Society of Limnology (SIL), Berlin, 09.08.2022.
July 2021, new publication:
Morón-Asensio, R., Schuler, D., Wiedlroither, A., Offterdinger, M., Kurmayer, R., 2021. Differential Labeling of Chemically Modified Peptides and Lipids among Cyanobacteria Planktothrix and Microcystis. Microorganisms 9:1578. https://doi.org/10.3390/microorganisms10040695
May 2021, presentations:
Morón-Asensio, R. co-authors: Kurmayer, R.: Monitoring of intracellular and extracellular toxin/bioactive peptide production under physiological stress conditions through labeling by chemical modification. 12th International Conference on Toxic Cyanobacteria (ICTC), Toledo, Ohio, 24.05.2022.
Morón-Asensio, R. co-authors: Kurmayer, R.: Differential labeling of chemically modified Cyanotoxins and Lipids among cyanobacteria Planktothrix and Microcystis. SETAC Europe 31st Annual Meeting: Global challenges - An Emergency for Environmental Sciences, Brüssel (online), 05.05.2021.
February 2020, new publication:
Kurmayer, R., E. Entfellner, T. Weisse, M. Offterdinger, A. Rentmeister & L. Deng, 2020. Chemically labeled toxins or bioactive peptides show a heterogeneous intracellular distribution and low spatial overlap with autofluorescence in bloom-forming cyanobacteria. Scientific Reports 10(1):2781, https://www.nature.com/articles/s41598-020-59381-w
February 2020:
Rubén Morón Asensio started his PhD within the SLOT project. A warm welcome on board!
December 2019:
David Schuler started his work as Lab technician within the SLOT project. A warm welcome on board!
November 2019:
Christoph Kotzorek started his MSc thesis project within the SLOT project. A warm welcome on board!
June 2019, presentation:
Kurmayer, R.: Localization of Microcystin and Anabaenopeptin Peptides on a Subcellular Level in the Cyanobacteria Planktothrix and Microcystis by Means of Noninvasive Peptide Labelling. Gordon Research Conference on Mycotoxins and Phycotoxins, Easton, 17.06.2019.
Personnel & Contacts
Associate Professor and Head of the group of Molecular ecology and physiology of cyanobacteria.
PhD student project SLOT. Molecular ecology and physiology of cyanobacteria.
Laboratory technician in Molecular ecology and physiology of cyanobacteria.
Daniel Holzer
MSc student project SLOT
Christoph Kotzorek
MSc student project SLOT
Kristin Kuntz
MSc student project SLOT
Kathrin Geir
Student coworker
Publications
Kurmayer R., Morón Asensio R. (2024). Real-Time Observation of Clickable Cyanotoxin Synthesis in Bloom-Forming Cyanobacteria Microcystis aeruginosa and Planktothrix agardhii. Toxins 16(12):526. doi:10.3390/toxins16120526
Kurmayer, R. (2024). Imaging Data management plan according to the FWF guideline: Original images are archived in long-term OA archives according to the Creative Commons License CC BY 4.0 (PDF)
Kurmayer R., Entfellner E., Weisse T., Offterdinger M., Rentmeister A., Deng L. (2020). Chemically labeled toxins or bioactive peptides show a heterogeneous intracellular distribution and low spatial overlap with autofluorescence in bloom-forming cyanobacteria. Sci Rep 10, 2781, https://doi.org/10.1038/s41598-020-59381-w
Rott E., Kurmayer R., Holzinger A., Sanders D.G. (2021). Contrasting endolithic habitats for cyanobacteria in spring calcites of the European Alps. Nova Hedwigia 112(1-2): 17-48. doi: 10.1127/nova_hedwigia/2021/0615
Morón-Asensio R., Schuler D., Wiedlroither A., Offterdinger M., Kurmayer R. (2021). Differential Labeling of Chemically Modified Peptides and Lipids among Cyanobacteria Planktothrix and Microcystis. Microorganisms 9(8):1578. doi:10.3390/microorganisms9081578
Morón-Asensio, R., D. Schuler, A. Wiedlroither, M. Offterdinger & R. Kurmayer, (2022). Correction: Morón-Asensio et al. Differential Labeling of Chemically Modified Peptides and Lipids among Cyanobacteria Planktothrix and Microcystis. Microorganisms 2021, 9, 1578. Microorganisms 10(4):695.
Entfellner E., Li R., Jiang Y., Ru J., Blom J., Deng L., Kurmayer R. (2022). Toxic/bioactive peptide synthesis genes rearranged by insertion sequence elements among the bloom-forming cyanobacteria Planktothrix. Front Microbiol doi:10.3389/fmicb.2022.901762
Olokotum M., Humbert J.-F., Quiblier C., Okello W., Semyalo R., Troussellier M., Marie B., Baumann K., Kurmayer R., Bernard C. (2022). Characterization of Potential Threats from Cyanobacterial Toxins in Lake Victoria Embayments and during Water Treatment. Toxins 14(10):664, https://doi.org/10.3390/toxins14100664
Entfellner, E., Baumann K.B.L., Edwards C., Kurmayer R. (2023). 'High Structural Diversity of Aeruginosins in Bloom-Forming Cyanobacteria of the Genus Planktothrix as a Consequence of Multiple Recombination Events', Marine drugs 21: 638. https://doi.org/10.3390/md21120638
Purker, M., Dobrovolny S. , Kreuml M. , Hufnagl P. , Indra A. , Kurmayer R., (2023). Quantitative relationships among high-throughput sequencing, cyanobacteria toxigenic genotype abundance and microcystin occurrence in bathing waters, Science of The Total Environment 901:165934 https://doi.org/10.1016/j.scitotenv.2023.165934
Jablonska, M., Cerasino L., Boscaini A., Capelli C., Greco C., Klemenčič A.K., Mischke U., Salmaso N., Kurmayer R. (2024). Distribution of toxigenic cyanobacteria in Alpine lakes and rivers as revealed by molecular screening. Water Research 121783, https://doi.org/10.1016/j.watres.2024.121783
