Chemistry and Physics of super cooled water and ice
Our research focuses on understanding the fascinating properties of amorphous systems, which lack long-range order in their atomic or molecular structure. We investigate the physics and chemistry of phase transitions in these materials, such as the glass-to-liquid transition and crystallization kinetics, which depend on factors such as temperature, pressure, and the presence of crystalline seeds.
We also study the different phases of ice, including metastable and stable phases, and surface premelting, which can have important implications for the behavior of water and ice in various environments.
Another important area of our research is low-temperature chemistry, which involves studying chemical reactions and intermediates at very low temperatures. We use techniques such as hyperquenching to rapidly cool samples to very low temperatures, allowing us to trap and study highly reactive intermediates that are difficult to isolate under normal conditions. Our work on low-temperature chemistry includes the isolation of metastable intermediates like H2CO3.
Through our research, we aim to deepen our understanding of the properties and behavior of these fascinating systems, and to apply this knowledge to a range of fields, from materials science to atmospheric chemistry.

Assoc.-Prof. Dr. Mag. Thomas Loerting

We warmly welcome Felix Münzker, our new Master student. He will work on IR transmission of ice.
March 2026
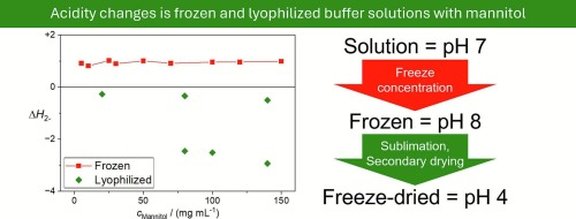
New publication in International Journal of Pharmaceutics: Acidity changes in glycine and L-histidine buffers, mannitol, and their mixtures after freezing and lyophilization
Freezing and lyophilization are commonly used methods for stabilizing pharmaceutical and biochemical formulations. However, these processes can introduce a variety of freezing-induced stresses that may lead not to stabilization, but rather to the destabilization of active molecules. In this study, we investigate the effects of cooling rate, initial pH, mannitol concentration, and lyophilization on acidity in glycine and L-histidine buffer systems in the solid state (both frozen and lyophilized) using UV–VIS spectroscopy and differential scanning calorimetry. The findings may inform the rational design of more robust stabilization strategies.
Link to the publication
February 2026
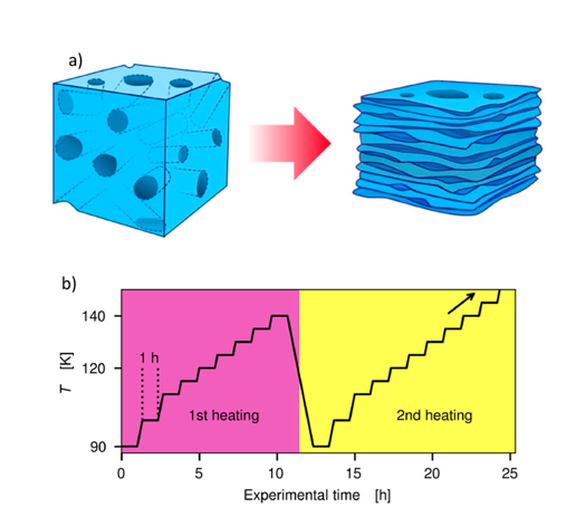
We are pleased to announce our latest publication in The Journal of Chemical Physics: Pore-collapse in amorphous solid water: A dynamics study
Vapor-deposited amorphous ice, so-called amorphous solid water, exhibits complex structural and morphological transformations upon heating. A network of micropores, present at the deposition temperature (80 K), collapses at 100–145 K, and a glass transition takes place simultaneously above 120 K. Here, we separate the two processes by allowing the micropores to collapse upon heating, which is monitored by small-angle x-ray scattering experiments.
Link to the publication
January 2026

We are pleased to welcome our new PhD students Mahdiyeh Moradi and Ammu Ammu Parakkattukudy Vijayan, as well as our new Master student Ythan Shaw. They all joined in the past month.
November 2025
Professor Thomas Loerting, accompanied by his team members recently made an exciting visit to Hamburg for crucial measurements at DESY.
Kontakt
Institut für Physikalische Chemie
Josef-Möller-Haus
Innrain 52c
A - 6020 Innsbruck
Österreich