Nanostructured Model Catalysis
Our group focuses on the mechanistic understanding of processes at the solid/gas interface on materials relevant for reactions in sustainable catalysis, such as methanol steam or methane dry reforming and the selective catalytic reduction of nitrogen oxide. Materials range from oxide over metal-oxide systems to intermetallic compounds and alloys and by combining model systems under ultra-high vacuum and (powder) materials under technologically relevant conditions we are devoted to close the “pressure” and “materials” gap in catalysis. Our approach is the exclusive use of in situ and operando structural and spectroscopic techniques to study the catalyst behavior under close-to-real conditions.
This interdisciplinary approach involves collaboration between researchers from various fields, including materials science, chemistry, physics, and engineering. We strive to develop innovative solutions to complex scientific problems and contribute to the advancement of the field.
NEWS
New Article Published in Small
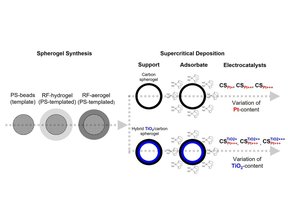
How can supporting design and controlled nanoparticle deposition improve electrocatalyst activity and durability?
We demonstrate that immobilizing Pt nanoparticles on nanoporous carbon spherogels with tunable TiO₂ sublayers enables precise control over particle size, dispersion, and spacing. The resulting catalysts show enhanced hydrogen evolution performance and superior stability compared to commercial Pt/C, with minimal degradation and suppressed nanoparticle growth. This approach highlights how engineered support morphology and metal–support interactions can deliver highly active and durable next-generation electrocatalysts.
https://onlinelibrary.wiley.com/doi/full/10.1002/smll.202512049
New Members (2025/2026)
We warmly welcome our new PhD student, Olena Polozhiy, and our new Master’s student, Benjamin Pichler. Both have joined our group in recent months, and we look forward to working with them.
New Article Published in JACS
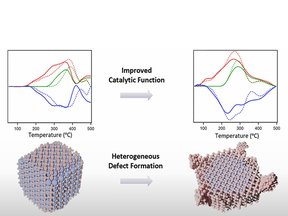
How can nanoscale defect engineering enhance catalytic activity in perovskites?
Using aberration-corrected electron microscopy and spectroscopy, we show that A-site-deficient La0.7Fe0.7Mn0.3O3 forms ultrathin (2–3 unit cell) defective surface layers that transition into a stoichiometric bulk. These layers stabilize FeOx nanoparticles (∼1–several nm), which act as active sites for NO reduction by CO via the Mars–van Krevelen mechanism. This work demonstrates how controlled A-site nonstoichiometry creates active interfaces, offering a strategy to design more efficient redox catalysts.
New Article Published in ACS Catalysis
How does a Pd/Zr catalyst truly behave under the demanding conditions of high-conversion DRM,and what governs its carbon fate?
By coupling temperature-resolved reaction profiling with XPS, SEM, and EDX, this study deciphers the fundamental pathways of carbon formation and regeneration, revealing how feed composition, conversion level, and regeneration atmosphere dictate coke evolution and removal. In comparison to Ni/Zr, it highlights the decisive role of accessible metal–oxide phase boundaries in CO₂ activation and catalyst recovery.
But ultimately, how can these insights be harnessed to design noble metal systems that sustain both high activity and effective carbon control?
New Article Published in JACS
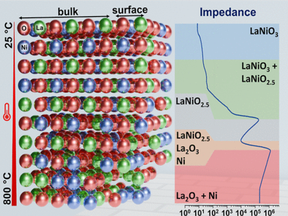
How can the hidden structural and chemical transformations of LaNiO₃ during redox cycling be revealed in real time?
By integrating in situ impedance spectroscopy with XRD, XPS, and thermoanalysis, this work deciphers bulk and surface transitions, from oxygen-deficient phases to full decomposition into La₂O₃ and Ni, while capturing defect dynamics, redox reversibility, and conductivity evolution.
But how does impedance, as a noninvasive probe, achieve such exceptional sensitivity to both surface reorganization and deep structural change?
New Article Published in ACS Catalysis
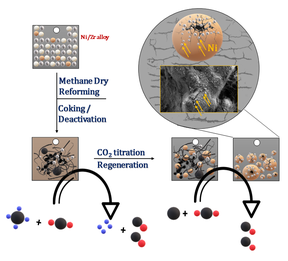
How can a coked Ni–Zr catalyst not only be restored, but transformed into a more active state?
Under DRM conditions, Ni–Zr evolves into a Ni–ZrO₂ interfacial phase prone to progressive coking. Yet, through intermediate regeneration with pure CO₂ via the inverse Boudouard reaction, coke is efficiently removed while simultaneously inducing Ni redispersion and optimizing metal–oxide interfaces.
But how does CO₂-driven regeneration unlock a catalyst state that surpasses its original activity?
Intermetallic-Based Materials for Methane Dry Reforming (FWF WEAVE PIN1004125. 03/26-03/92)
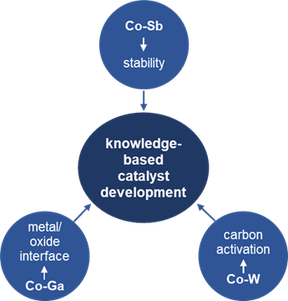
The dry reforming of methane (DRM) is a viable heterogeneously catalyzed reaction to convert two harmful greenhouse gases carbon dioxide and methane into syngas (hydrogen and carbon monoxide) that can be further used to produce synthetic fuels. Much effort has been placed into the development of catalyst materials to overcome the coking and sintering limitations of the archetypical Ni-based catalysts. Several material classes have been screened and a common denominator of all systems is the highly dynamic nature of the catalytically active sites that must enable a full reversible methane and carbon dioxide activation-and-release cycle. The reactivity of carbon, due to reactant activation, is therefore crucial to obtain a highly active material. Intermetallic compound-based materials have evolved as particular promising catalyst materials in a variety of reactions due to their outstanding structural and electronic properties, including dry reforming of methane. A leading theme underscoring the highly dynamic nature is the use of intermetallic compounds as precursor materials to in situ form corresponding metal-oxide systems in either the dry reforming mixture or by pre-reduction in hydrogen. These metal-oxide systems then serve as the connector to other more common dry reforming catalysts. This steered decomposition pathway has been developed mostly on Zr-based intermetallic compounds. As such, studies on intermetallic materials mostly rely on materials that can be decomposed, but studies on stable intermetallic compounds are virtually non-existent. To fill this knowledge gap, the project provides a systematic assessment of the catalytic properties of intermetallic compounds in the dry reforming of methane. By comparing a set of cobalt-based intermetallic compounds in the cobalt-antimony, cobalt-gallium and cobalt-tungsten phase diagram, we span the entire stability region of DRM-stable (cobalt-antimony), DRM-unstable (cobalt-gallium, formation of a cobalt-gallium oxide interface) and carbide-forming (cobalt-tungsten) intermetallic compounds. This allows studying and comparing the intrinsic DRM properties of stable intermetallic compounds to those of a metal-oxide composite resulting from in situ decomposition and the reactivity of carbon within the same set of intermetallic compounds using a common leading element. Consequently, this approach enables addressing the teamwork between the different catalytically active components of intermetallic compounds and facilitates the establishment of reliable structure-property relationships in the dry reforming of methane.
If you are interested, please send your application to simon.penner@uibk.ac.at
In situ studies of catalyst deactivation and regeneration in methane dry reforming (ReMade@ARI 34204. 12/24-12/26)
By using Ni-Zr and Pd-Zr alloy catalyst precursors for methane dry reforming (DRM) applications, we study the initial activation, deactivation, and regeneration of the catalyst material by a combination of in situ high-resolution electron microscopy (HR-TEM), electron-energy loss spectroscopy (EELS), energy-dispersive X-ray spectroscopy (EDXS), and secondary electron imaging on a Hitachi 5000 electron microscope. Preliminary catalytic and structural studies by ex situ SEM have revealed that the initial Ni-Zr and Pd-Zr alloys transform into Ni-ZrO2 and Pd-ZrO2 with carbon filament formation, successively deactivating the catalyst by Ni and Pd particle encapsulation after several catalytic cycles. For Ni-Zr, we have preliminarily shown that regeneration by a CO2 treatment is possible via the inverse Boudouard reaction (CO2+C ↔ 2CO), which leads to carbon removal, partial encapsulation of the larger Ni particles by ZrO2 layers, corrosion of larger Ni particles into smaller ones with enhanced coking resistance and, in turn, restoration and even improvement of catalytic DRM activity. The results prove that regeneration of coked catalysts by CO2 is possible, but the structural prerequisites are yet largely unclear. To shed light on the structural transformations of the Ni-Zr alloys in the respective deactivation and regeneration steps, we want to focus on the proposed in situ experiments, especially in the regeneration step. To use the Hitachi 5000 microscope, it is necessary to FIB-cut lamellae parallel to the surface of the support material, as the Ni-Zr and Pd-Zr alloys are prepared on a Zr foil. Catalyst regeneration by CO2 is a two-fold improvement in catalyst improvement: it avoids oxidative regeneration in oxygen, which often leads to particle sintering and provides an efficient additional means of re-using the harmful greenhouse gas carbon dioxide.
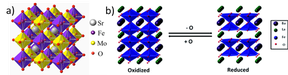
Double Perovskites as Mixed Ionic-Electronic Conductors (FWF EINZEL P 35770. 09/22-10/26)
The main focus of the project “Double Perovskite-based Mixed Ionic-Electronic Conductors (MIEC)” lies on the synthesis and understanding of a new prospective class of double perovskite systems on strontium-iron-vanadium/niobium basis (Sr2Fe1+x(V/Nb)1-xO6-d) with controlled defect chemistry, and their in situ and operando characterization with respect to their use as prospective MIEC materials in solid oxide cells. Double perovskites belong to the class of complex oxides, which - due to their manifold physico-chemical properties – exhibit the potential to substitute to date used conventional materials with unfavorable behavior under extreme experimental conditions in fuel cell systems. To do so, the usual main obstacle of poor structural stability under relevant technological operating conditions has to be overcome. The focus of the project is the direct correlation of in situ/operando-determined redox chemistry and structural stability with catalytic properties in internal reforming reactions. The most important step hereby is the knowledge-based synthesis of the materials under controlled oxygen fugacity to directly steer the defect concentration and the redox behavior. In this respect, we will directly exploit the physico-chemical properties of the constituting elements to influence and trigger favorable material’s properties. Of equal and paramount importance is the structural and (electro-) chemical characterization of the materials under in situ and operando conditions, i.e. in the state of operation.
To achieve these goals, we utilize a portfolio of characterization methods capable of being operated under such close-to-real technological conditions. This includes in situ/operando structural characterization (X-ray diffraction, electron microscopy), electrochemical performance (electrochemical impedance spectroscopy), surface chemical characterization (FT-Infrared Spectroscopy), monitoring the electronic structure (in situ X-ray photoelectron spectroscopy) and quantitatively assessing the defect concentration. The scientific and methodological approach allows us to gain unprecedented insight into the structure-property relationships of double-perovskite materials in the working state of the materials and to optimize generalized synthesis routines to prospective new catalytic materials for SOC operation.
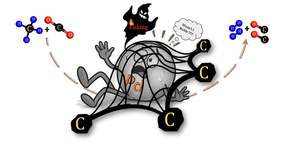
Optimization of Carbon Chemistry in Methane Dry Reforming (FWF EINZEL P 36926-N. 03/23-07/27)
The main goal of the project “Optimization of the Carbon Chemistry in Methane Dry Reforming” is connected to the in situ/ operando experimental assessment and fundamental understanding of reactive carbon intermediates in different branches of the methane dry reforming network. In depth understanding of carbon reactivity and coke suppression will enable a focused, knowledge-based design of methane dry reforming. The central focus of the project is the direct correlation of the in situ/ operando determined reactivity of different carbon species with catalytic methane dry reforming activity. By exploiting the controlled in situ decomposition of selected intermetallic compounds towards active metal-oxide interfaces, we are able to quantify the reactivity of carbon bound in variable oxidation states between carbides and oxy-carbonates, including the suppression and re-mobilization of coke. The influence of material-specific redox chemistry, involving the the elementary carbon-forming and -converting reactions within the dry reforming network, will be scrutinized. To extract detailed structure-reactivity relationships, we will complement kinetic reactivity studies with in situ/ operando bulk and surface characterization under realistic reaction conditions. To accomplish these tasks, we rely on an exclusive portfolio of in situ and operando characterization methods capable of operation under close-to-technological conditions. This includes in situ/operando structural (X-ray diffraction, electron microscopy, Raman spectroscopy) and surface chemical characterization (X-ray photoelectron spectroscopy and FT-Infrared Spectroscopy), as well as integral quantification of carbon (thermo-gravimetry). We complement this characterization with kinetic reactor studies of important reactions within the dry reforming network and with theoretical assessment of the observed carbon reactivity trends. The controlled in situ decomposition of variable intermetallic precursors allows to induce a broad range of interfacial and bulk carbon species. Our methodological approach will provide novel material-specific insights into viable carbon reaction pathways within the methane dry reforming network.
Contact
Institut für Physikalische Chemie
Josef-Möller-Haus
Innrain 52c
A - 6020 Innsbruck
Österreich
Teamleaders
Assoz. Prof. Dr.
Bernhard Klötzer
Priv.Doz- Dr.
Simon Penner